| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
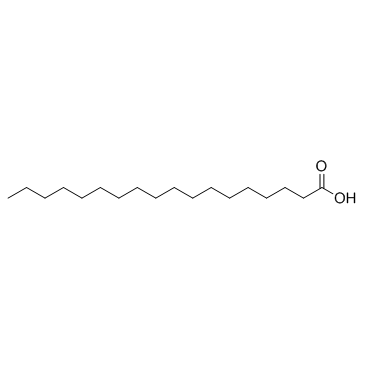 |
硬脂酸
CAS:57-11-4 |
|
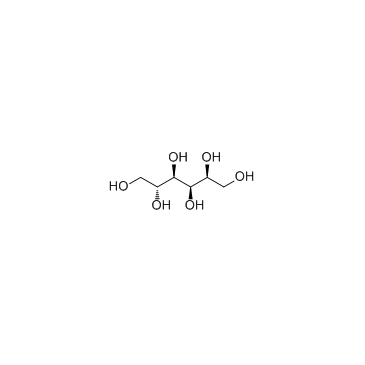 |
山梨醇
CAS:50-70-4 |
|
 |
甲醇
CAS:67-56-1 |
|
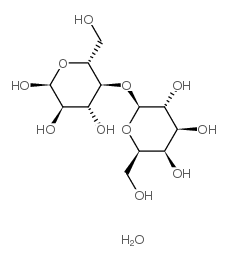 |
D-(+)-乳糖-水合物
CAS:64044-51-5 |
|
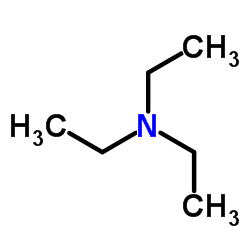 |
三乙胺
CAS:121-44-8 |