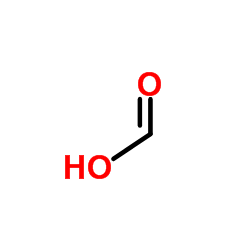
甲酸锂
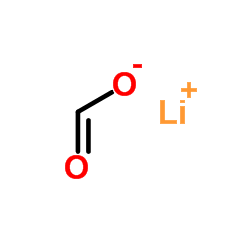
甲酸锂结构式

|
常用名 | 甲酸锂 | 英文名 | Lithium formate |
|---|---|---|---|---|
| CAS号 | 556-63-8 | 分子量 | 69.974 | |
| 密度 | 1.46g/cm3 | 沸点 | 100.8ºC | |
| 分子式 | CH3LiO3 | 熔点 | 8.4ºC | |
| MSDS | N/A | 闪点 | 29.9ºC | |
| 符号 |

GHS07 |
信号词 | Warning |
| 中文名 | 甲酸锂 |
|---|---|
| 英文名 | lithium formate |
| 英文别名 | 更多 |
| 密度 | 1.46g/cm3 |
|---|---|
| 沸点 | 100.8ºC |
| 熔点 | 8.4ºC |
| 分子式 | CH3LiO3 |
| 分子量 | 69.974 |
| 闪点 | 29.9ºC |
| 精确质量 | 70.024223 |
| PSA | 40.13000 |
| InChIKey | XKPJKVVZOOEMPK-UHFFFAOYSA-M |
| SMILES | O=C[O-].[Li+] |
| 外观性状 | 白色结晶状粉末 |
| 储存条件 | 室温 |
| 计算化学 | 1.疏水参数计算参考值(XlogP):无 2.氢键供体数量:0 3.氢键受体数量:2 4.可旋转化学键数量:0 5.互变异构体数量:无 6.拓扑分子极性表面积:40.1 7.重原子数量:4 8.表面电荷:0 9.复杂度:13.5 10.同位素原子数量:0 11.确定原子立构中心数量:0 12.不确定原子立构中心数量:0 13.确定化学键立构中心数量:0 14.不确定化学键立构中心数量:0 15.共价键单元数量:2 |
| 符号 |

GHS07 |
|---|---|
| 信号词 | Warning |
| 危害声明 | H319-H336 |
| 警示性声明 | P280-P304 + P340 + P312-P305 + P351 + P338-P337 + P313 |
| 危害码 (欧洲) | Xi |
| 风险声明 (欧洲) | 36/37/38 |
| 安全声明 (欧洲) | 26-36/37/39 |
| 危险品运输编码 | UN1219 - class 3 - PG 2 - Isopropanol, solution |
|
~95% 
甲酸锂 556-63-8 |
| 文献:Jana, Anukul; Tavcar, Gasper; Roesky, Herbert W.; John, Michael Dalton Transactions, 2010 , vol. 39, # 40 p. 9487 - 9489 |
|
~% 
甲酸锂 556-63-8 |
| 文献:Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy, , vol. 66, # 3 p. 754 - 760 |
|
~% 
甲酸锂 556-63-8 |
| 文献:Angewandte Chemie - International Edition, , vol. 50, # 28 p. 6411 - 6414 |
|
~% 
甲酸锂 556-63-8 |
| 文献:Journal of the Indian Chemical Society, , vol. 67, # 1 p. 19 - 22 |
|
~% 
甲酸锂 556-63-8
详细
|
| 文献:J. Gen. Chem. USSR (Engl. Transl.), , vol. 59, # 3.2 p. 687 - 691,607 - 610 |
|
~% 
甲酸锂 556-63-8
详细
|
| 文献:J. Gen. Chem. USSR (Engl. Transl.), , vol. 59, # 3.2 p. 687 - 691,607 - 610 |
|
~% 
甲酸锂 556-63-8 |
| 文献:DE381957 ; Fortschr. Teerfarbenfabr. Verw. Industriezweige, vol. 14, p. 241 |
|
~% 
甲酸锂 556-63-8
详细
|
| 文献:J. Gen. Chem. USSR (Engl. Transl.), , vol. 51, # 2 p. 299 - 302,239 - 242 |
|
~% 
甲酸锂 556-63-8
详细
|
| 文献:J. Gen. Chem. USSR (Engl. Transl.), , vol. 51, # 2 p. 299 - 302,239 - 242 |
| 甲酸锂上游产品 9 | |
|---|---|
| 甲酸锂下游产品 10 | |
|
Bionanowhiskers from jute: preparation and characterization.
Carbohydr. Polym. 92(2) , 1116-23, (2013) Bionanowhiskers were extracted from jute by acid hydrolysis. At first cellulose microfibrils were formed by alkali treatment. Addition of an acid to the microfibrils triggered the formation of cellulo... |
|
|
Monoclonal antibodies selective for α-synuclein oligomers/protofibrils recognize brain pathology in Lewy body disorders and α-synuclein transgenic mice with the disease-causing A30P mutation.
J. Neurochem. 126(1) , 131-44, (2013) Inclusions of intraneuronal alpha-synuclein (α-synuclein) can be detected in brains of patients with Parkinson's disease and dementia with Lewy bodies. The aggregation of α-synuclein is a central feat... |
|
|
Density functional theory study on the adsorption and decomposition of the formic acid catalyzed by highly active mushroom-like Au@Pd@Pt tri-metallic nanoparticles.
Phys. Chem. Chem. Phys. 15(13) , 4625-33, (2013) Local structures and adsorption energies of a formic acid molecule and its decomposed intermediates (H, O, OH, CO, HCOO, and COOH) on highly electrocatalytically active mushroom-like Au-core@Pd-shell@... |
| Ameisensaeure,Lithiumformiat |
| formic acid,lithium formate |
| Formic acid, lithium salt, hydrate (1:1:1) |
| Lithiummethanoat |
| Lithiumformiatemonohydrate |
| Lithium formate hydrate (1:1:1) |
| lithium methanoate |
| Lithiumformiat |
| formic acid Li salt |
| Formic acid, lithium salt |
| Formic acid, lithium salt (1:1) |
| Formic acid lithium |
| Lithiumformiate-1-hydrate |
| Lithium formate |
| Einecs 209-133-0 |
| FORMIC ACID LITHIUM SALT |


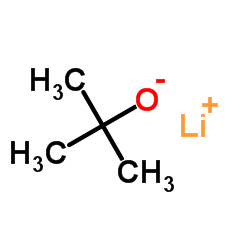
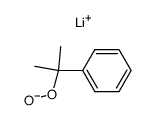
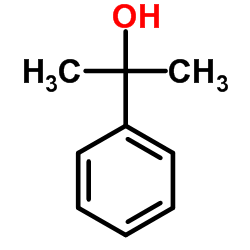
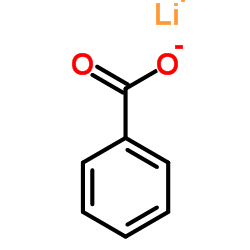
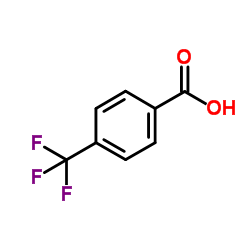 CAS号455-24-3
CAS号455-24-3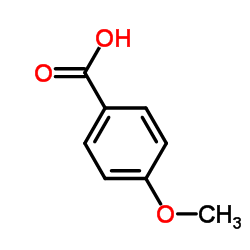 CAS号100-09-4
CAS号100-09-4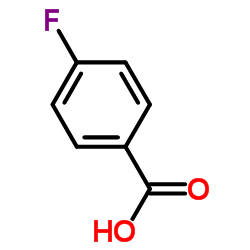 CAS号456-22-4
CAS号456-22-4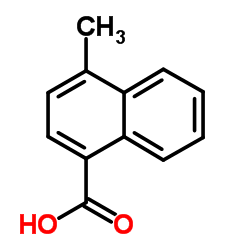 CAS号4488-40-8
CAS号4488-40-8 CAS号445-29-4
CAS号445-29-4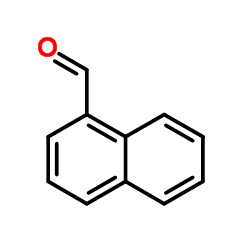 CAS号66-77-3
CAS号66-77-3 CAS号92-92-2
CAS号92-92-2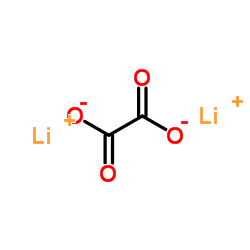 CAS号553-91-3
CAS号553-91-3 CAS号67-56-1
CAS号67-56-1







