泼尼卡酯
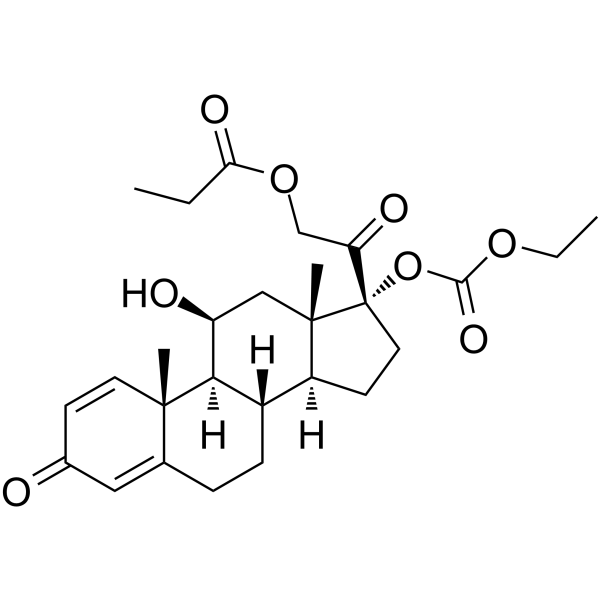
泼尼卡酯结构式

|
常用名 | 泼尼卡酯 | 英文名 | Prednicarbate |
|---|---|---|---|---|
| CAS号 | 73771-04-7 | 分子量 | 488.570 | |
| 密度 | 1.3±0.1 g/cm3 | 沸点 | 640.7±55.0 °C at 760 mmHg | |
| 分子式 | C27H36O8 | 熔点 | 110 - 112ºC | |
| MSDS | N/A | 闪点 | 209.4±25.0 °C |
泼尼卡酯用途Prednicarbate是一种外用皮质类固醇药物。Prednicarbate可用于炎症性皮肤病的研究,如特应性皮炎[1][2]。 |
| 中文名 | 泼尼卡松 |
|---|---|
| 英文名 | [2-(17-ethoxycarbonyloxy-11-hydroxy-10,13-dimethyl-3-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-17-yl)-2-oxo-ethyl] propanoate |
| 中文别名 | 11Β,17,21-三羟基孕甾-1,4-二烯-3,20二酮-17-(乙基碳酸酯)-21-丙酸酯 | 泼尼卡酯 |
| 英文别名 | 更多 |
| 描述 | Prednicarbate是一种外用皮质类固醇药物。Prednicarbate可用于炎症性皮肤病的研究,如特应性皮炎[1][2]。 |
|---|---|
| 相关类别 | |
| 体外研究 | Prednicarbate(0.25%;每天涂抹在耳朵皮肤部位2天)可恢复2,4-二硝基氯苯(DNCB)诱导的小鼠特应性皮炎[2]。 |
| 参考文献 |
| 密度 | 1.3±0.1 g/cm3 |
|---|---|
| 沸点 | 640.7±55.0 °C at 760 mmHg |
| 熔点 | 110 - 112ºC |
| 分子式 | C27H36O8 |
| 分子量 | 488.570 |
| 闪点 | 209.4±25.0 °C |
| 精确质量 | 488.241028 |
| PSA | 116.20000 |
| LogP | 4.02 |
| InChIKey | FNPXMHRZILFCKX-KAJVQRHHSA-N |
| SMILES | CCOC(=O)OC1(C(=O)COC(=O)CC)CCC2C3CCC4=CC(=O)C=CC4(C)C3C(O)CC21C |
| 外观性状 | 白色晶体粉末 |
| 蒸汽压 | 0.0±4.3 mmHg at 25°C |
| 折射率 | 1.560 |
| 分子结构 | 1、 摩尔折射率:125.65 2、 摩尔体积(cm3/mol):388.5 3、 等张比容(90.2K):1044.6 4、 表面张力(dyne/cm):52.2 5、 极化率(10-24cm3):49.81 |
| 危险品运输编码 | NONH for all modes of transport |
|---|
|
~30% 
泼尼卡酯 73771-04-7 |
| 文献:Stache; Fritsch; Rupp; Hitzel; Fehlhaber Arzneimittel-Forschung/Drug Research, 1985 , vol. 35, # 12 p. 1753 - 1757 |
|
~% 
泼尼卡酯 73771-04-7 |
| 文献:Arzneimittel-Forschung/Drug Research, , vol. 35, # 12 p. 1753 - 1757 |
|
~% 
泼尼卡酯 73771-04-7 |
| 文献:Arzneimittel-Forschung/Drug Research, , vol. 35, # 12 p. 1753 - 1757 |
|
~% 
泼尼卡酯 73771-04-7 |
| 文献:Arzneimittel-Forschung/Drug Research, , vol. 35, # 12 p. 1753 - 1757 |
以泼尼松龙prednisolone为原料。
110g(0.305mo1)泼尼松龙溶于2.2L无水二氧六烷,加入147ml(0.70mo1)原碳酸四乙酯和12g对甲苯磺酸。在22℃搅拌或放置17h。将反应液倾入17L水中,用碳酸氢钠中和。首先析出一油状物,放置1~24h后形成结晶。过滤,水洗。在五氧化二磷存在下,于高真空干燥至恒重,得126.5g化合物(Ⅰ),收率90%。用二氯甲烷-乙醇-乙醚重结晶,得到可用于分析的化合物(Ⅰ),熔点155℃。
125g(0.27mo1)化合物(Ⅰ)溶于1.4L无水乙酸,在18℃和搅拌下,加入10ml水。在18℃放置5h后,将其和含有1kg氯化钠的18L水一起搅拌。过滤收集沉淀,水洗。在五氧化二磷存在下,高真空干燥至恒重,得72g粗产品。滤液减压浓缩,又析出固体,干燥后得36g。两者合计108g化合物(Ⅱ)的粗品,收率92.3%。该粗品用无酸的二氯甲烷-乙醇重结晶后,即可用于下步反应。用于分析的标准品可用硅胶层析,无水二氯甲烷洗脱,得精品,熔点145℃。
从化合物(Ⅱ)可有两种方法得到泼尼卡酯。
方法1:68.5g(0.158mo1)化合物(Ⅱ)溶于680ml无水吡啶,在0℃和搅拌下,60min内,滴加18.2g(0.197mo1)丙酰氯。在0℃搅拌30min后,再在20℃搅拌2h。将反应液倾入含有1kg氯化钠的18L水中。过滤收集沉淀,用水彻底洗涤。在五氧化二磷存在下,高真空干燥至恒重,得63g泼尼卡酯粗品,收率81.4%。10g该粗品用200g硅胶层析,甲苯-乙酸乙酯洗脱。浓缩后用乙醇-乙醚重结晶,得5.7g泼尼卡酯精品,收率57%,熔点110~112℃。4g该精品在80ml异丙醚中加热60min,过滤,干燥后得熔点更高的结晶,熔点183℃。
方法2:1.05kg(2.43mo1)化合物(Ⅱ)的粗品溶于10L无水二氯甲烷在20℃滴加0.336I。(2.6mo1)丙酸酐,然后加入50g(0.41mo1)4-二甲氨基吡啶。在20℃搅拌16h后,反应液依次用水、碳酸氢钠水溶液和水洗,用无水硫酸钠干燥。减压蒸去溶剂,剩余物和上述方法相似,层析后再用异丙醚重结晶,得360g泼尼卡酯粗品,收率30.4%,熔点110~112℃。
|
Cetuximab-induced skin exanthema: prophylactic and reactive skin therapy are equally effective.
J. Cancer Res. Clin. Oncol. 139(10) , 1667-72, (2013) Treatment with cetuximab is accompanied by the development of an acneiform follicular skin exanthema in more than 80 % of patients. Severe exanthema (grade III/IV) develops in about 9-19 % of patients... |
|
|
Drug release and skin penetration from solid lipid nanoparticles and a base cream: a systematic approach from a comparison of three glucocorticoids.
Skin Pharmacol. Physiol. 24(4) , 199-209, (2011) Solid lipid nanoparticles (SLNs) can enhance drug penetration into the skin, yet the mechanism of the improved transport is not known in full. To unravel the influence of the drug-particle interaction... |
|
|
Efficacy of crisis intervention treatment with topical corticosteroid prednicarbat with and without partial wet-wrap dressing in atopic dermatitis.
Dermatology 212(1) , 66-9, (2006) The wet-wrap treatment has been reported to be beneficial in acute episodes of atopic dermatitis (AD) skin lesions.The efficacy of topical corticosteroid prednicarbat with and without additional wet-w... |
|
实验名称:Luminescence-based cell-based primary high throughput screening assay to identify ago...
来源:The Scripps Research Institute Molecular Screening Center
靶标:mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id:OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
实验名称:Fluorescence-based cell-based primary high throughput screening assay to identify ago...
来源:The Scripps Research Institute Molecular Screening Center
靶标:muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id:CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
实验名称:Dicer-mediated maturation of pre-microRNA
来源:Center for Chemical Genomics, University of Michigan
靶标:N/A
External Id:TargetID_659_CEMA
|
|
实验名称:Fluorescence-based cell-based primary high throughput screening assay to identify pos...
来源:The Scripps Research Institute Molecular Screening Center
靶标:muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id:CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
实验名称:Fluorescence polarization-based biochemical high throughput primary assay to identify...
来源:The Scripps Research Institute Molecular Screening Center
靶标:RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id:SIAE_INH_FP_1536_1X%INH PRUN
|
|
实验名称:MITF Measured in Cell-Based System Using Plate Reader - 2084-01_Activator_SinglePoint...
来源:Broad Institute
靶标:N/A
External Id:2084-01_Activator_SinglePoint_HTS_Activity
|
|
实验名称:Counterscreen for inhibitors of the fructose-bisphosphate aldolase (FBA) of M. tuberc...
来源:The Scripps Research Institute Molecular Screening Center
靶标:N/A
External Id:GDH-TPI_INH_ABS_1536_1X%INH CSRUN
|
|
实验名称:qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
来源:NCGC
External Id:HERG01
|
|
实验名称:Inhibition of cell viability relative to arbidol control (inhibition index > 1 indica...
来源:ChEMBL
靶标:Severe acute respiratory syndrome coronavirus 2
External Id:CHEMBL4303819
|
| (11b)-17-[(Ethoxycarbonyl)oxy]-11-hydroxy-21-(1-oxopropoxy)pregna-1,4-diene-3,20-dione |
| (11β)-17-[(Ethoxycarbonyl)oxy]-11-hydroxy-3,20-dioxopregna-1,4-dien-21-yl propionate |
| propanoate de 2-{(8S,9S,10R,11S,13S,14S,17R)-17-[(éthoxycarbonyl)oxy]-11-hydroxy-10,13-diméthyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodécahydro-3H-cyclopenta[a]phénanthrén-17-yl}-2-oxoéthyle |
| 11b,17,21-Trihydroxypregna-1,4-diene-3,20-dione 17-(Ethyl Carbonate) 21-Propionate |
| 2-{(8S,9S,10R,11S,13S,14S,17R)-17-[(Ethoxycarbonyl)oxy]-11-hydroxy-10,13-dimethyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-17-yl}-2-oxoethylpropanoat |
| Dermatop |
| 11β,17,21-Trihydroxypregna-1,4-diene-3,20-dione 17-(ethyl carbonate) 21-propionate |
| 2-{(8S,9S,10R,11S,13S,14S,17R)-17-[(ethoxycarbonyl)oxy]-11-hydroxy-10,13-dimethyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-17-yl}-2-oxoethyl propanoate |
| (11β)-17-[(ethoxycarbonyl)oxy]-11-hydroxy-3,20-dioxopregna-1,4-dien-21-yl propanoate |
| Carbonic acid, ethyl (11β)-11-hydroxy-3,20-dioxo-21-(1-oxopropoxy)pregna-1,4-dien-17-yl ester |
| Prednicarbate (200 mg) |
| Regemt |
| Prednicarbate |
| HOE-777 |
| S 77 0777 |
| Prednisolone 17-(ethyl carbonate) 21-propionate |
| Regenit |
| Prednitop |
| S-77-0777 |
| Prednisolone 17-Ethylcarbonate 21-Propionate |


 CAS号5740-62-5
CAS号5740-62-5





