| Structure | Name/CAS No. | Articles |
|---|---|---|
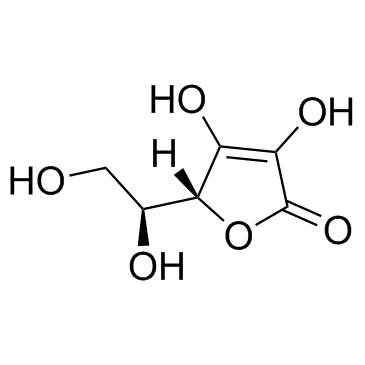 |
Ascorbic acid
CAS:50-81-7 |
|
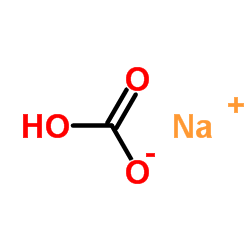 |
SodiuM bicarbonate
CAS:144-55-8 |
|
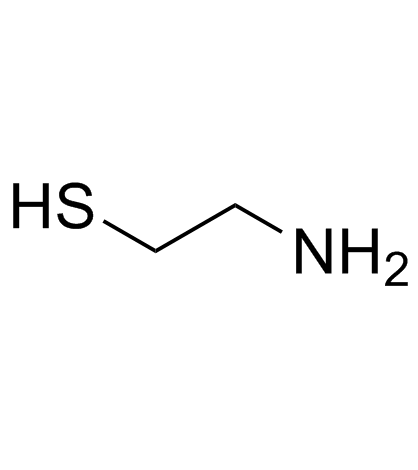 |
2-Aminoethanethiol
CAS:60-23-1 |
|
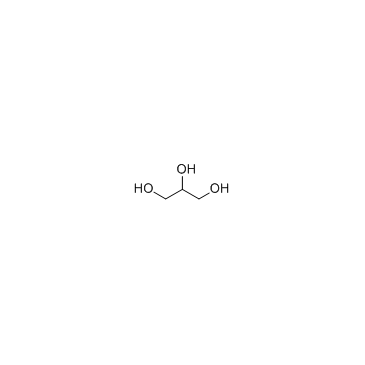 |
Glycerol
CAS:56-81-5 |
|
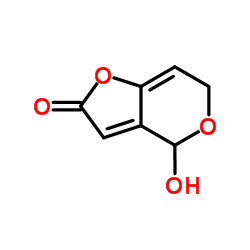 |
Glucose oxidase
CAS:9001-37-0 |