| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
5-Methoxy-1H-indole-3-carbaldehyde
CAS:10601-19-1 |
|
![Imidazo[1,2-a]pyridine Structure](https://image.chemsrc.com/caspic/372/274-76-0.png) |
Imidazo[1,2-a]pyridine
CAS:274-76-0 |
|
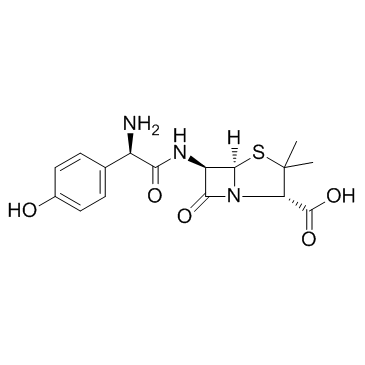 |
Amoxicillin
CAS:26787-78-0 |