Imidazo[1,2-a]pyridine
![Imidazo[1,2-a]pyridine Structure](https://image.chemsrc.com/caspic/372/274-76-0.png)
Imidazo[1,2-a]pyridine structure
|
Common Name | Imidazo[1,2-a]pyridine | ||
|---|---|---|---|---|
| CAS Number | 274-76-0 | Molecular Weight | 118.136 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 103ºC (1 mmHg) | |
| Molecular Formula | C7H6N2 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 113ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | Imidazo[1,2-a]pyridine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 103ºC (1 mmHg) |
| Molecular Formula | C7H6N2 |
| Molecular Weight | 118.136 |
| Flash Point | 113ºC |
| Exact Mass | 118.053101 |
| PSA | 17.30000 |
| LogP | 1.35 |
| Appearance of Characters | Powder | White to off-white |
| Index of Refraction | 1.628 |
| InChIKey | UTCSSFWDNNEEBH-UHFFFAOYSA-N |
| SMILES | c1ccn2ccnc2c1 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2933990090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Conversion of pyridine to imidazo[1,2-a]pyridines by copper-catalyzed aerobic dehydrogenative cyclization with oxime esters.
Org. Lett. 15(24) , 6254-7, (2013) A rapid and environmentally friendly conversion of pyridine to imidazo[1,2-a]pyridines has been developed via copper-catalyzed aerobic dehydrogenative cyclization with ketone oxime esters. |
|
|
One-pot three-component synthesis of 3-nitro-2-arylimidazo[1,2-a]pyridine derivatives using air as an oxidant.
Chem. Asian J. 7(9) , 2028-31, (2012)
|
|
|
Synthesis and biological evaluation of imidazo[1,2-a]pyridine derivatives as novel PI3 kinase p110alpha inhibitors.
Bioorg. Med. Chem. 15(1) , 403-12, (2007) 3-{1-[(4-Fluorophenyl)sulfonyl]-1H-pyrazol-3-yl}-2-methylimidazo[1,2-a]pyridine, 2a, was discovered in our chemical library as a novel p110alpha inhibitor with an IC(50) of 0.67microM, through screeni... |
|
Name: Selectivity index, ratio of CC50 for human HepG2 cells to IC50 for Leishmania donovan...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2444092
|
|
Name: Displacement of Y-27632 from ROCK-1 assessed as drop in intensity of binding signals ...
Source: ChEMBL
Target: Rho-associated protein kinase 1
External Id: CHEMBL1647663
|
|
Name: Displacement of [3H]-NECA from A2A receptor in rat striatal membrane in presence of A...
Source: ChEMBL
Target: Adenosine receptor A2a
External Id: CHEMBL4058588
|
|
Name: Displacement of [3H]-DPCPX from A1 receptor in rat whole brain membrane
Source: ChEMBL
Target: Adenosine receptor A1
External Id: CHEMBL4058587
|
|
Name: Dissociation constant, pKa of the compound
Source: ChEMBL
Target: N/A
External Id: CHEMBL863765
|
|
Name: Inhibition of FLT3 (unknown origin) using FAM-EPLYWSFPA as substrate at 20 uM preincu...
Source: ChEMBL
Target: Receptor-type tyrosine-protein kinase FLT3
External Id: CHEMBL3420605
|
|
Name: Partition coefficient, log P of the compound
Source: ChEMBL
Target: N/A
External Id: CHEMBL4415147
|
|
Name: Distribution coefficient, log D at pH 7.4
Source: ChEMBL
Target: N/A
External Id: CHEMBL863769
|
|
Name: Antimicrobial activity against Leishmania donovani MHOM/IN/00/DEVI promastigotes afte...
Source: ChEMBL
Target: Leishmania donovani
External Id: CHEMBL2444096
|
| 1H-imidazo[1,2-a]pyridine |
| imidazo[1,2-a]-pyridine |
| imidazo[2,1-b]pyridine |
| Imidazo[1,2-a]pyridine |
| MFCD00005553 |
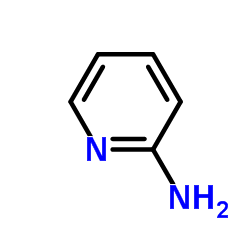 CAS#:504-29-0
CAS#:504-29-0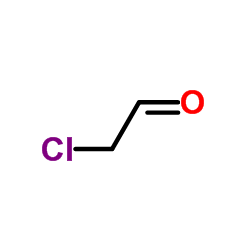 CAS#:107-20-0
CAS#:107-20-0![Imidazo[1,2-a]pyridine-3-carboxylic Acid Structure](https://image.chemsrc.com/caspic/054/6200-60-8.png) CAS#:6200-60-8
CAS#:6200-60-8 CAS#:1072-98-6
CAS#:1072-98-6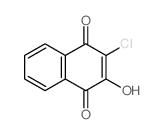 CAS#:1526-73-4
CAS#:1526-73-4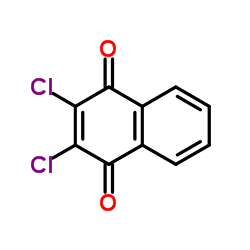 CAS#:117-80-6
CAS#:117-80-6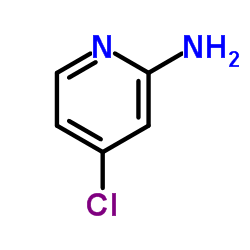 CAS#:19798-80-2
CAS#:19798-80-2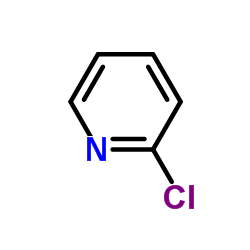 CAS#:109-09-1
CAS#:109-09-1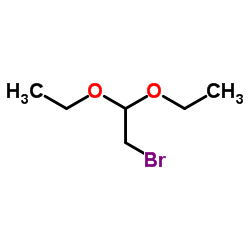 CAS#:2032-35-1
CAS#:2032-35-1 CAS#:6602-54-6
CAS#:6602-54-6![3-Nitroimidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/132/4926-45-8.png) CAS#:4926-45-8
CAS#:4926-45-8![3-Bromoimidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/046/4926-47-0.png) CAS#:4926-47-0
CAS#:4926-47-0![3-Iodoimidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/495/307503-19-1.png) CAS#:307503-19-1
CAS#:307503-19-1![3-(Chlorodifluoroacetyl)imidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/494/219296-24-9.png) CAS#:219296-24-9
CAS#:219296-24-9![Imidazo[1,2-a]pyridin-3-ylmethanol structure](https://image.chemsrc.com/caspic/460/30489-43-1.png) CAS#:30489-43-1
CAS#:30489-43-1![3-[(dimethylamino)methyl]imidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/310/2717-95-5.png) CAS#:2717-95-5
CAS#:2717-95-5![Ethanone, 1-imidazo[1,2-a]pyridin-3-yl structure](https://image.chemsrc.com/caspic/012/29096-64-8.png) CAS#:29096-64-8
CAS#:29096-64-8![3-(4-Chlorophenyl)-imidazo[1,2-a]pyridine structure](https://image.chemsrc.com/caspic/121/663946-08-5.png) CAS#:663946-08-5
CAS#:663946-08-5![Imidazo[1,2-a]pyridine-3-carbaldehyde structure](https://image.chemsrc.com/caspic/380/6188-43-8.png) CAS#:6188-43-8
CAS#:6188-43-8![Imidazo[1,2-a]pyridinium,1-methyl-, iodide (1:1) structure](https://image.chemsrc.com/caspic/299/15562-25-1.png) CAS#:15562-25-1
CAS#:15562-25-1
