| Structure | Name/CAS No. | Articles |
|---|---|---|
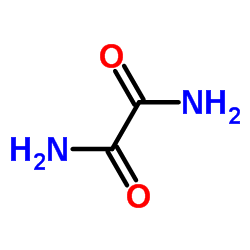 |
oxamide
CAS:471-46-5 |
|
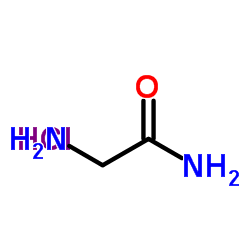 |
H-Gly-NH2.HCl
CAS:1668-10-6 |
|
 |
OXAMIC ACID
CAS:471-47-6 |