Clinical trials of topical corticosteroids in psoriasis: correlations with the vasoconstrictor assay.
R C Cornell
Index: Int. J. Dermatol. 31 Suppl 1 , 38-40, (1992)
Full Text: HTML
Abstract
It would be desirable to develop an alternative system to clinical studies to evaluate the potency of generic topical corticosteroids and of new formulations of existing innovator corticosteroids. The vasoconstrictor assay is a reliable method for testing potency; however, its results do not always agree with clinical studies. Psoriasis offers an ideal clinical model to evaluate corticoid potency because the ability to perform within-patient comparisons of the treatment of bilateral lesions permits meaningful comparisons with a relatively small sample size. The results of bilateral comparisons in psoriasis agreed with those of the vasoconstrictor assay in 20 of 23 comparisons of active agents and in numerous comparisons of active corticosteroids with a placebo. Eczematous dermatoses do not lend themselves well to bilateral paired comparison studies and therefore require parallel treatment studies with relatively large sample sizes to produce statistically significant comparisons.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
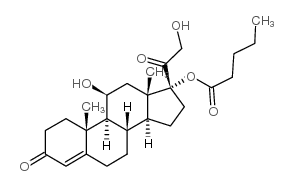 |
Hydrocortisone Valerate
CAS:57524-89-7 |
C26H38O6 |
|
Topical corticosteroid compounding: effects on physicochemic...
1989-11-01 [J. Am. Acad. Dermatol. 21(5 Pt 1) , 979-84, (1989)] |
|
Addition of a topically applied corticosteroid to a modified...
1985-11-01 [J. Am. Acad. Dermatol. 13(5 Pt 1) , 784-91, (1985)] |
|
Temporal infiltration of leukocyte subsets into mouse skin i...
1992-11-01 [Agents Actions 37(3-4) , 260-7, (1992)] |
|
Contact hypersensitivity to tixocortol pivalate.
1998-05-01 [J. Am. Acad. Dermatol. 38(5 Pt 1) , 691-5, (1998)] |
|
A dermatitis-eosinophilia syndrome. Treatment with methylpre...
1984-12-01 [Arch. Dermatol. 120(12) , 1595-7, (1984)] |