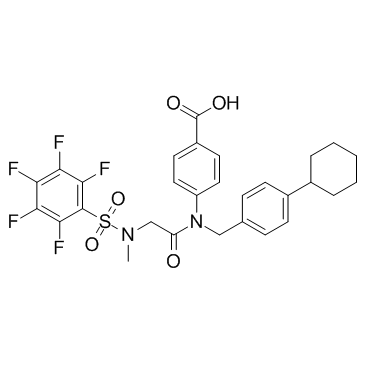
1456632-40-8
| Name | Benzoic acid, 4-[[(4-cyclohexylphenyl)methyl][2-[methyl[(2,3,4,5,6-pentafluorophenyl)sulfonyl]amino]acetyl]amino] |
|---|---|
| Synonyms |
4-[(4-Cyclohexylbenzyl){N-methyl-N-[(pentafluorophenyl)sulfonyl]glycyl}amino]benzoic acid
SH-4-54 4-[[(4-Cyclohexylphenyl)methyl][2-[methyl[(2,3,4,5,6-pentafluorophenyl)sulfonyl]amino]acetyl]amino]benzoic acid |
| Description | SH-4-54 is a most potent, small molecule, nonphosphorylated STAT inhibitor, with KDs of 300, 464 nM for STAT3 and STAT5, respectively. |
|---|---|
| Related Catalog | |
| Target |
STAT3:300 nM (Kd) STAT5:464 nM (Kd) |
| In Vitro | SH-4-54 potently kills glioblastoma brain cancer stem cells (BTSCs) and effectively suppresses STAT3 phosphorylation and its downstream transcriptional targets at low nM concentrations.SH-4-54 shows unprecedented cytotoxicity in human BTSCs, displays no toxicity in human fetal astrocytes, potently suppresses pSTAT3 with nanomolar IC50s, inhibiting STAT3's downstream targets, and shows no discernible off-target effects at therapeutic doses[1]. |
| In Vivo | SH-4-54 exhibits blood-brain barrier permeability potently controls glioma tumor growth, and inhibits pSTAT3 in vivo. SH-4-54 demonstrates the power of STAT3 inhibitors for the treatment of BTSCs and validates the therapeutic efficacy of a STAT3 inhibitor for GBM clinical application.SH-4-54 decreases pSTAT3 expression in tumor cells of treated mice. SH-4-54 appears to decrease proliferation and increase apoptosis of treated tumors[1]. |
| References |
| Density | 1.431±0.06 g/cm3 |
|---|---|
| Boiling Point | 717.2±70.0 °C at 760 mmHg |
| Molecular Formula | C29H27F5N2O5S |
| Molecular Weight | 610.592 |
| Flash Point | 387.5±35.7 °C |
| Exact Mass | 610.156067 |
| PSA | 103.37000 |
| LogP | 7.64 |
| Vapour Pressure | 0.0±2.4 mmHg at 25°C |
| Index of Refraction | 1.590 |
| Storage condition | -20℃ |
| Water Solubility | Insuluble (2.6E-5 g/L) (25 ºC) |