21947-98-8
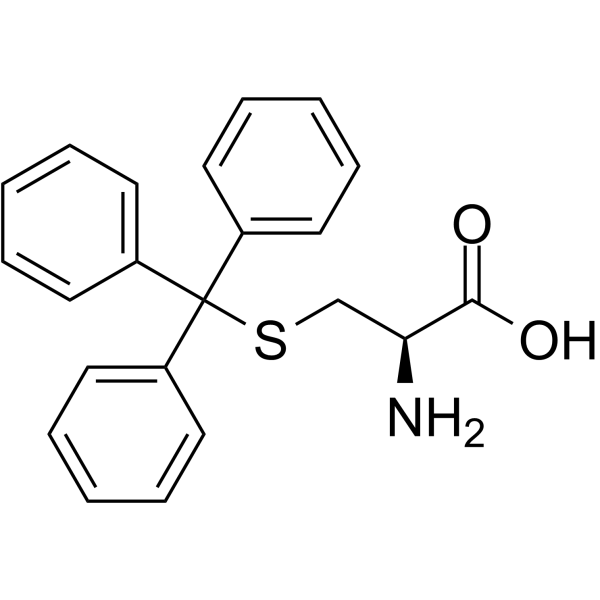
| Name | (2R)-2-[(2-methylpropan-2-yl)oxycarbonylamino]-3-tritylsulfanylpropanoic acid |
|---|---|
| Synonyms |
MFCD00038251
(R)-2-(tert-Butoxycarbonylamino)-3-(tritylthio)propanoic acid N-[(1,1-dimethylethoxy)carbonyl]-S-trityl-L-cysteine n-(tert-butoxycarbonyl)-s-trityl-l-cysteine S-trityl-N-Boc-(R)-cysteine N-{[(2-Methyl-2-propanyl)oxy]carbonyl}-S-trityl-L-cysteine N-t-butyloxycarbonyl-S-trityl-L-cysteine S-trityl-N-Boc-cysteine N-t-butoxycarbonyl-S-trityl-L-Cysteine Boc-S-trityl-L-cysteine Boc-Cys(Trt)-OH EINECS 244-674-6 N-Boc-S-trityl-L-cysteine |
| Description | (R)-2-((tert-Butoxycarbonyl)amino)-3-(tritylthio)propanoic acid is a cysteine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 610.9±55.0 °C at 760 mmHg |
| Melting Point | 143-146 °C(lit.) |
| Molecular Formula | C27H29NO4S |
| Molecular Weight | 463.589 |
| Flash Point | 323.3±31.5 °C |
| Exact Mass | 463.181732 |
| PSA | 100.93000 |
| LogP | 7.52 |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.596 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
|
~89% 
21947-98-8 |
| Literature: Venkateswara Rao, Boddu; Dhokale, Snehal; Rajamohanan, Pattuparambil R.; Hotha, Srinivas Chemical Communications, 2013 , vol. 49, # 92 p. 10808 - 10810 |
|
~% 
21947-98-8 |
| Literature: Organic Letters, , vol. 7, # 13 p. 2615 - 2618 |
|
~% 
21947-98-8 |
| Literature: European Journal of Medicinal Chemistry, , vol. 50, p. 383 - 392 |
|
~% 
21947-98-8 |
| Literature: Chemische Berichte, , vol. 102, p. 1048 - 1052 |
|
~% 
21947-98-8 |
| Literature: Canadian Journal of Chemistry, , vol. 57, p. 1388 - 1396 |
| Precursor 7 | |
|---|---|
| DownStream 4 | |