114297-20-0
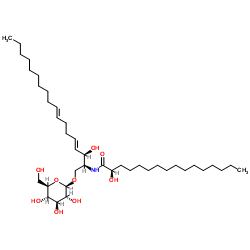
| Name | (2R)-2-hydroxy-N-[(3R,4E,8E)-3-hydroxy-1-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoctadeca-4,8-dien-2-yl]hexadecanamide |
|---|---|
| Synonyms |
(2R)-N-[(2S,3R,4E,8E)-1-(β-D-Glucopyranosyloxy)-3-hydroxy-4,8-octadecadien-2-yl]-2-hydroxyhexadecanamide
Soya-cerebroside I |
| Description | Soyacerebroside I is a glycosphingolipid, together with Soyacerebroside II, which can be isolated from the seeds of Glycine max Merrill (Leguminosae). Soyacerebroside II shows ionophoretic activity for Ca2+[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 870.1±65.0 °C at 760 mmHg |
| Molecular Formula | C40H75NO9 |
| Molecular Weight | 714.02 |
| Flash Point | 480.0±34.3 °C |
| Exact Mass | 713.544189 |
| PSA | 168.94000 |
| LogP | 10.07 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.530 |
| Hazard Codes | Xi |
|---|