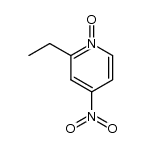
536-33-4
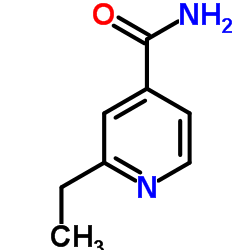
| Name | ethionamide |
|---|---|
| Synonyms |
2-ethyl-4-thiopyridylamide
2-ethyl-4-thiocarbamoylpyridine EINECS 208-628-9 Ethionamide 2-Ethyl-4-pyridinecarbothioamide Etionamid 2-Ethyl-4-pyridinecarbothioamide,Ethionamide Etioniamid Trecator Ethyonomide Thioamide Ethioniamide Ethinamide Ethylisothiamide 2-Ethylpyridine-4-carbothioamide 2-ethyl-4-thiocarbamoyl-4-pyridine MFCD00057361 Trecator-SC 2-Ethylthioisonicotinamide |
| Description | Ethionamide(2-ethylthioisonicotinamide) is an antibiotic used in the treatment of tuberculosis.Target: AntibacterialEthionamide is a second-line antitubercular agent that inhibits mycolic acid synthesis. It also may be used for treatment of leprosy. Ethionamide is a prodrug. It is activated by the enzyme EthA, a mono-oxygenase in Mycobacterium tuberculosis, and binds NAD+ to form an adduct which inhibits InhA in the same way as isoniazid. Expression of the ethA gene is controlled by EthR, a transcriptional repressor. It is understood that improving ethA expression will increase the efficacy of ethionamide and so EthR inhibitors are of great interest to co-drug developers. The action may be through disruption of mycolic acid [1, 2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.17 g/cm3 |
|---|---|
| Boiling Point | 167 °C / 1mmHg |
| Melting Point | 164 °C |
| Molecular Formula | C8H10N2S |
| Molecular Weight | 166.24300 |
| Flash Point | 133.2ºC |
| Exact Mass | 166.05600 |
| PSA | 71.00000 |
| LogP | 1.97850 |
| Index of Refraction | 1.599 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H361 |
| Precautionary Statements | P281 |
| Personal Protective Equipment | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22;R63 |
| Safety Phrases | S36/37 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NS0350000 |
| HS Code | 2933990090 |
|
~80% 
536-33-4 |
| Literature: Bergman, Jan; Pettersson, Birgitta; Hasimbegovic, Vedran; Svensson, Per H. Journal of Organic Chemistry, 2011 , vol. 76, # 6 p. 1546 - 1553 |
|
~% 
536-33-4 |
| Literature: Bulletin de la Societe Chimique de France, , p. 687,691 |
|
~% 
536-33-4 |
| Literature: Bulletin de la Societe Chimique de France, , p. 687,691 |
|
~% 
536-33-4 |
| Literature: Bulletin de la Societe Chimique de France, , p. 687,691 |
|
~% 
536-33-4 |
| Literature: Zhurnal Obshchei Khimii, , vol. 29, p. 915,918; engl. Ausg. S. 898 |
|
~% 
536-33-4 |
| Literature: Zhurnal Obshchei Khimii, , vol. 29, p. 915,918; engl. Ausg. S. 898 |
|
~% 
536-33-4 |
| Literature: Zhurnal Obshchei Khimii, , vol. 29, p. 915,918; engl. Ausg. S. 898 |
|
~% 
536-33-4 |
| Literature: Zhurnal Obshchei Khimii, , vol. 29, p. 915,918; engl. Ausg. S. 898 |
| Precursor 8 | |
|---|---|
| DownStream 2 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |