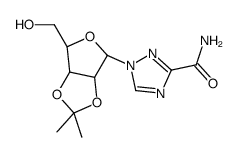
36791-04-5
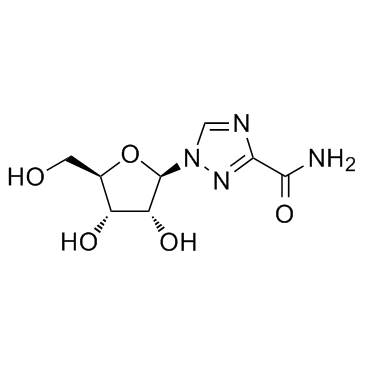
| Name | ribavirin |
|---|---|
| Synonyms |
1-(b-D-ribofuranosyl)-1H-1,2,4-triazole-3-carboxamide
RTC 1-β-D-ribofuranosyl-1,2,4-triazole-3-carboxamide Ribavirin 1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-1H-1,2,4-triazole-3-carboxamide UNII-49717AWG6K MFCD00058564 rtca Rebetol Ravanex 1-(β-D-Ribofuranosyl)-1H-1,2,4-triazole-3-carboxamide Virazide Virazole VIRAZID Copegus varazid 1-β-D-ribofuranosyl-1H-1,2,4-triazole-3-carboxamide tribavirin 1-(β-D-Ribofuranosyl)-1,2,4-triazole-3-carboxamide virazol Viramid 1-b-D-Ribofuranosyl-1H-1,2,4-triazole-3-carboxamide vilona |
| Description | Ribavirin is an antiviral agent against a broad spectrum of viruses including HCV, HIVl, and RSV. |
|---|---|
| Related Catalog | |
| In Vitro | Ribavirin (5, 10 and 20 μM) does not produce any significant effect on neither cell viability nor nitrites level in non-stimulated microglia. Treatmentof LPS-stimulated microglia with 5, 10 and 20 μM Ribavirin induces reduction of NO2 levels for 43% (p<0.05), 53% (p<0.05) and 59% (p<0.05), respectively. Ribavirin (10 mM) insignificantly decreases the cell surface area in non-stimulated culture, but significantly reduces cell surface area (by 32%, p<0.05) in LPS-stimulated microglia[3]. Ribavirin is active against DENV, with an EC50 of 3 μM in A549 cells, and combination of CM-10-18 with ribavirin demonstrates a clear enhancement in the reduction of virus replication[4]. |
| In Vivo | ALT, AST activities and bilirubin levels are significantly loared by administration of JAT in combination with interferon and ribavirin (p<0.01). JAT, interferon or ribavirin alone with CCl4, livers appear to exhibit some liver protection against CCl4 as evident by the presence of normal hepatic cords, absence of necrosis and lesser fatty infiltration. Groups treated with JAT, Peg-interferon and ribavirin separately or in combination shows reduction in the expression of TGF- β and Bax. In the group treated by triple combination of interferon, ribavirin, and JAT, the expression level of p53 is markedly reduced[1]. Ribavirin capsules (400 mg of ribavirin)-treated Wistar rats show a significant decrease in activin-A and significant increase in follistatin at the serum and liver levels. Ribavirin has strong antiviral activity only when ribavirin is combined with either IFN-α or Peg-IFN-α[2]. Ribavirin (40 mg/kg, p.o.) significantly improves the antiviral efficacy of CM-10-18 in mice. Ribavirin inhibits DENV virus infection in cultured cells, but it is ineffective in reducing viremia in monotherapy[4]. |
| Cell Assay | The effect of Ribavirin on microglial cell viability is evaluated by the sulforhodamine B (SRB) chemosensitivity assay. Briefly, LPS-stimulated microglial cells are incubated for 48 h in the presence or absence of Ribavirin. Afterward, the cells are fixed in 10% (w/v) trichloroacetic acid for 1 h at 4°C, rinsed in tap water and stained with 0.4% (w/v) SRB in 1% acetic acid (100 μL/well) for 30 min at room temperature (RT). The cells are then rinsed three times in 1% acetic acid to remove the unbound stain. The protein bound stain is extracted with 200 μL 10 mM Tris base (pH 10.5) per well. The optical density is read at 540 nm, with correction at 670 nm. The results are presented as percentage of the control (non-stimulated/untreated microglial cells), that is arbitrarily set to 100%. |
| Animal Admin | The in vivo efficacy experiments are largely performed, using a dengue viremia model in AG129 mouse (defective of both type I and type II interferon receptors). Each experiment contains 6 mice (7 to 8 week-old) per dosing group. The mice are challenged with DENV (serotype 2, TSV01 strain), at 5×106 pfu/mouse via i.p. injection. Imino sugars are dosed twice daily at 12 hr intervals, and ribavirin is dosed once daily, for 3 consecutive days post-infection. Blood samples are drawn 3 days post-infection for determination of plasma virus titer by plaque assay. |
| References |
| Density | 2.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 639.8±65.0 °C at 760 mmHg |
| Melting Point | 174-176°C |
| Molecular Formula | C8H12N4O5 |
| Molecular Weight | 244.205 |
| Flash Point | 340.7±34.3 °C |
| Exact Mass | 244.080765 |
| PSA | 143.72000 |
| LogP | -2.26 |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.823 |
| Storage condition | 2-8°C |
| Water Solubility | >=10 G/100 ML AT 19 ºC |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H360 |
| Precautionary Statements | P201-P280-P308 + P313 |
| Personal Protective Equipment | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | Xi |
| Risk Phrases | R61 |
| Safety Phrases | 53-22-45-37/39-26 |
| RIDADR | UN 3152 9/PG 2 |
| WGK Germany | 3 |
| RTECS | XZ4250000 |
| Packaging Group | II |
| Hazard Class | 9 |
| HS Code | 2933290090 |
| Precursor 8 | |
|---|---|
| DownStream 8 | |
| HS Code | 2933290090 |
|---|---|
| Summary | 2933290090. other compounds containing an unfused imidazole ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |


![[(2R,3R,4R,5R)-3,4-diacetyloxy-5-(3-cyano-1,2,4-triazol-1-yl)oxolan-2-yl]methyl acetate structure](https://image.chemsrc.com/caspic/248/40371-99-1.png)

![[(2R,3R,4R,5R)-5-(3-carbamoyl-1,2,4-triazol-1-yl)-3,4-dihydroxy-oxolan-2-yl]methoxyphosphonic acid structure](https://image.chemsrc.com/caspic/170/40925-28-8.png)