ADRA1D receptor antagonist 1 free base
Modify Date: 2025-08-26 20:28:28
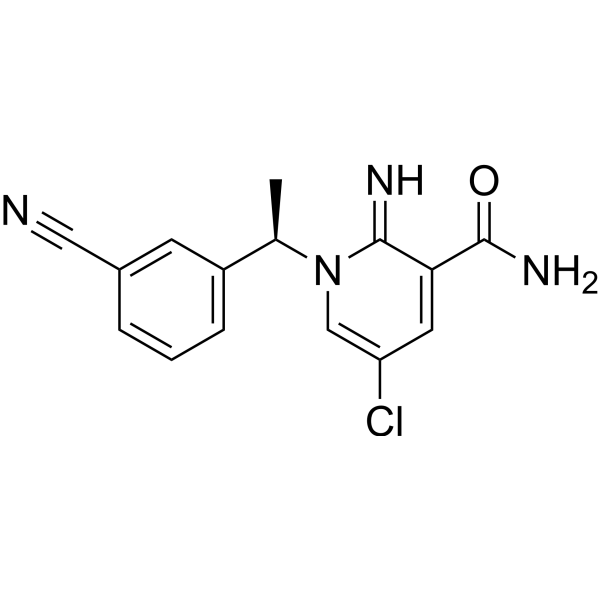
ADRA1D receptor antagonist 1 free base structure
|
Common Name | ADRA1D receptor antagonist 1 free base | ||
|---|---|---|---|---|
| CAS Number | 1191908-24-3 | Molecular Weight | 300.74 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C15H13ClN4O | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of ADRA1D receptor antagonist 1 free baseADRA1D receptor antagonist 1 (free base) (compound (R)-9s) is an orally active, potent and selective human α1D-adrenoceptor (α1D-AR) antagonist (Ki=1.6 nM). ADRA1D receptor antagonist 1 (free base) dose-dependently inhibits bladder contraction with an IC30 value of 15 nM. ADRA1D receptor antagonist 1 (free base) can be used in studies of overactive bladder disorders such as urinary urgency, frequency and incontinence. |
| Name | ADRA1D receptor antagonist 1 free base |
|---|
| Description | ADRA1D receptor antagonist 1 (free base) (compound (R)-9s) is an orally active, potent and selective human α1D-adrenoceptor (α1D-AR) antagonist (Ki=1.6 nM). ADRA1D receptor antagonist 1 (free base) dose-dependently inhibits bladder contraction with an IC30 value of 15 nM. ADRA1D receptor antagonist 1 (free base) can be used in studies of overactive bladder disorders such as urinary urgency, frequency and incontinence. |
|---|---|
| Related Catalog | |
| In Vivo | ADRA1D receptor antagonist 1 (free base) (10 µg/kg; p.o.; single) inhibits cyclophosphamide-induced urinary frequency in rats[1]. ADRA1D receptor antagonist 1 (free base) (4.4 µg/kg; i.v.; single) inhibits bladder contraction with an IC 30 value of 15 nM in rats[1]. Animal Model: Rats with cyclophosphamide-induced cystitis[1]. Dosage: 10 µg/kg Administration: Oral administration; single Result: Increased voiding intervals. Animal Model: Rats with BOO (bladder outlet obstruction)[1]. Dosage: 4.4 µg/kg Administration: Intravenous injection; single Result: Dose-dependently decreased the non-voiding bladder contractions during urinary storage phase. |
| Molecular Formula | C15H13ClN4O |
|---|---|
| Molecular Weight | 300.74 |
| InChIKey | CDJPHTNUARAVMH-SECBINFHSA-N |
| SMILES | CC(c1cccc(C#N)c1)n1cc(Cl)cc(C(N)=O)c1=N |
|
Name: Redox activity H2DCFDA assay (redox-free)
Source: ChEMBL
Target: N/A
External Id: CHEMBL4880078
|
|
Name: Redox activity Resazurin assay (0.1 µM compound)
Source: ChEMBL
Target: N/A
External Id: CHEMBL4880077
|
|
Name: Redox activity Resazurin assay (1 µM compound)
Source: ChEMBL
Target: N/A
External Id: CHEMBL4880076
|
|
Name: Redox activity Resazurin assay (10 µM compound)
Source: ChEMBL
Target: N/A
External Id: CHEMBL4880075
|
|
Name: Redox activity H2DCFDA assay (100 µM TCEP)
Source: ChEMBL
Target: N/A
External Id: CHEMBL4880079
|
|
Name: 5-HT1 Eurofins-Panlabs ligand profiling
Source: ChEMBL
Target: 5-hydroxytryptamine receptor 1F
External Id: CHEMBL4883179
|
|
Name: beta3 Eurofins-Panlabs ligand profiling
Source: ChEMBL
Target: Beta-3 adrenergic receptor
External Id: CHEMBL4883178
|
|
Name: beta2 Eurofins-Panlabs ligand profiling
Source: ChEMBL
Target: Beta-2 adrenergic receptor
External Id: CHEMBL4883177
|
|
Name: beta1 Eurofins-Panlabs ligand profiling
Source: ChEMBL
Target: Beta-1 adrenergic receptor
External Id: CHEMBL4883176
|
|
Name: KinomeScan assay: inhibition of ERK8
Source: ChEMBL
Target: Mitogen-activated protein kinase 15
External Id: CHEMBL4879707
|
Total 113, Current Page 1 of 12
1
2
3
4
5