Sodium Urate
Modify Date: 2024-01-02 19:30:47
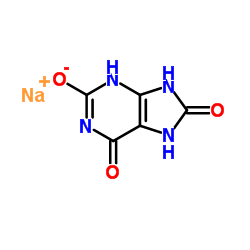
Sodium Urate structure
|
Common Name | Sodium Urate | ||
|---|---|---|---|---|
| CAS Number | 1198-77-2 | Molecular Weight | 190.092 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C5H3N4NaO3 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | N/A | |
Use of Sodium UrateUric acid sodium, scavenger of oxygen radical, is a very important antioxidant that help maintains the stability of blood pressure and antioxidant stress. Uric acid sodium can remove reactive oxygen species (ROS) such as singlet oxygen and peroxynitrite, inhibiting lipid peroxidation[1][2]. |
| Name | uric acid sodium salt |
|---|---|
| Synonym | More Synonyms |
| Description | Uric acid sodium, scavenger of oxygen radical, is a very important antioxidant that help maintains the stability of blood pressure and antioxidant stress. Uric acid sodium can remove reactive oxygen species (ROS) such as singlet oxygen and peroxynitrite, inhibiting lipid peroxidation[1][2]. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | Uric acid (400 μM; 48 hours) protects Caco-2 cells from indomethacin-induced lipid peroxidation[2]. Co-treatment of cells with Indomethacin and Uric acid (200 μM IND plus 400 μM UA; 24 hours) significantly decreases ROS levels compared to those in cells incubated with indomethacin alone. Cell viability in Caco-2 cells treated with both Indomethacin and Uric acid (200 μM IND plus 400 μM UA; 24 hours) is higher than that in cells treated with indomethacin alone. Uric acid has a protective effect on indomethacin-induced intestinal cell changes through its antioxidant activity[2]. |
| In Vivo | Uric acid (250 mg/kg; p.o.) ameliorates indomethacin-induced enteropathy[2]. Oral administration of uric acid decreased ROS accumulation in the ileum in a mouse model of indomethacin-induced enteropathy[2]. Animal Model: Eight-week-old male C57BL/6J mice[2] Dosage: 250 mg/kg body weight Administration: P.o. Result: When mice treated with indomethacin were concurrently administered uric acid orally, ulcer areas were significantly reduced, in a uric acid dose-dependent manner. |
| References |
| Molecular Formula | C5H3N4NaO3 |
|---|---|
| Molecular Weight | 190.092 |
| Exact Mass | 190.010284 |
| PSA | 117.46000 |
| Stability | Stable. |
|
Section 1. Chemical Product and Company Identification Uric Acid, Sodium Salt Common Name/ Trade Name Uric Acid, Sodium Salt Section 4. First Aid Measures Check for and remove any contact lenses. In case of contact, immediately flush eyes with plenty of water for at least
Eye Contact 15 minutes. WARM water MUST be used. Get medical attention. In case of contact, immediately flush skin with plenty of water. Cover the irritated skin with an emollient. Remove Skin Contact contaminated clothing and shoes. Wash clothing before reuse. Thoroughly clean shoes before reuse. Get medical attention. Serious Skin ContactNot available. InhalationIf inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical attention. Serious InhalationNot available. Do NOT induce vomiting unless directed to do so by medical personnel. Never give anything by mouth to an Ingestion unconscious person. If large quantities of this material are swallowed, call a physician immediately. Loosen tight clothing such as a collar, tie, belt or waistband. Serious IngestionNot available. Section 5. Fire and Explosion Data Flammability of the Product May be combustible at high temperature. Auto-Ignition Temperature Not available. Not available. Flash Points Not available. Flammable Limits These products are carbon oxides (CO, CO2), nitrogen oxides (NO, NO2...). Products of Combustion Fire Hazards in Presence of Slightly flammable to flammable in presence of heat. Various Substances Risks of explosion of the product in presence of mechanical impact: Not available. Explosion Hazards in Risks of explosion of the product in presence of static discharge: Not available. Presence of Various Substances SMALL FIRE: Use DRY chemical powder. Fire Fighting Media LARGE FIRE: Use water spray, fog or foam. Do not use water jet. and Instructions Material in powder form, capable of creating a dust explosion. As with most organic solids, fire is possible at elevated Special Remarks on temperatures Fire Hazards Fine dust dispersed in air in sufficient concentrations, and in the presence of an ignition source is a potential dust Special Remarks on explosion hazard. Explosion Hazards Section 6. Accidental Release Measures Use appropriate tools to put the spilled solid in a convenient waste disposal container. Finish cleaning by spreading Small Spill water on the contaminated surface and dispose of according to local and regional authority requirements. Use a shovel to put the material into a convenient waste disposal container. Finish cleaning by spreading water on Large Spill the contaminated surface and allow to evacuate through the sanitary system. Uric Acid, Sodium Salt Section 7. Handling and Storage Keep away from heat. Keep away from sources of ignition. Ground all equipment containing material. Do not Precautions breathe dust. Avoid contact with eyes. Wear suitable protective clothing. If you feel unwell, seek medical attention and show the label when possible. Keep away from incompatibles such as oxidizing agents. Keep container tightly closed. Keep container in a cool, well-ventilated area. Storage Section 8. Exposure Controls/Personal Protection Use process enclosures, local exhaust ventilation, or other engineering controls to keep airborne levels below Engineering Controls recommended exposure limits. If user operations generate dust, fume or mist, use ventilation to keep exposure to airborne contaminants below the exposure limit. Personal ProtectionSplash goggles. Lab coat. Dust respirator. Be sure to use an approved/certified respirator or equivalent. Gloves. Personal Protection in Case Splash goggles. Full suit. Dust respirator. Boots. Gloves. A self contained breathing apparatus should be used to avoid inhalation of the product. Suggested protective clothing might not be sufficient; consult a specialist BEFORE of a Large Spill handling this product. Not available. Exposure Limits Section 9. Physical and Chemical Properties Solid. (Crystalline powder.)Not available. Physical state andO dor appearance Not available. Taste 190.1 g/mole Molecular Weight White to yellowish. Color Not available. pH (1% soln/water) Not available. Boiling Point Not available. Melting Point Not available. Critical Temperature Not available. Specific Gravity Not applicable. Vapor Pressure Not available. Vapor Density Not available. Volatility Not available. Odor Threshold Not available. Water/Oil Dist. Coeff. Not available. Ionicity (in Water) Not available. Dispersion Properties Very slightly soluble in hot water. Solubility Section 10. Stability and Reactivity Data The product is stable. Stability Not available. Instability Temperature Conditions of Instability Excess heat, incompatible materials. Incompatibility with various Reactive with oxidizing agents. substances Not available. Corrosivity Uric Acid, Sodium Salt Not available. Special Remarks on Reactivity Not available. Special Remarks on Corrosivity Will not occur. Polymerization Section 11. Toxicological Information Inhalation. Ingestion. Routes of Entry LD50: Not available. Toxicity to Animals LC50: Not available. Chronic Effects on Humans Not available. Other Toxic Effects onSlightly hazardous in case of skin contact (irritant), of ingestion, of inhalation. Humans Not available. Special Remarks on Toxicity to Animals May cause adverse reproductive effects. Special Remarks on Chronic Effects on Humans Acute Potential Health Effects: Special Remarks on other Skin: May cause skin irritation. Toxic Effects on Humans Eyes: Causes eye irritation. Inhalation: May cause respiratory tract irritation. Ingestion: Expected to be a low hazard. Section 12. Ecological Information Not available. Ecotoxicity Not available. BOD5 and COD Products of Biodegradation Possibly hazardous short term degradation products are not likely. However, long term degradation products may arise. The product itself and its products of degradation are not toxic. Toxicity of the Products of Biodegradation Not available. Special Remarks on the Products of Biodegradation Section 13. Disposal Considerations Waste must be disposed of in accordance with federal, state and local environmental control Waste Disposal regulations. Section 14. Transport Information Not a DOT controlled material (United States). DO T Cl assi fi cati on Not applicable. Identification Not applicable. Special Provisions for Transport Uric Acid, Sodium Salt DO T (Pi ctograms) Section 15. Other Regulatory Information and Pictograms TSCA 8(b) inventory: Uric Acid, Sodium Salt Federal and State Regulations California prop. 65: This product contains the following ingredients for which the State of California has found to California cause cancer which would require a warning under the statute: No products were found. Proposition 65 Warnings California prop. 65: This product contains the following ingredients for which the State of California has found to cause birth defects which would require a warning under the statute: No products were found. Other RegulationsEINECS: This product is on the European Inventory of Existing Commercial Chemical Substances (EINECS No. 214-838-1). Canada: Listed on Canadian Non-Domestic Substance List (NDSL). China: Not listed on National Inventory. Japan: Not listed on National Inventory (ENCS). Korea: Not listed on National Inventory (KECI). Philippines: Not listed on National Inventory (PICCS). Australia: Not listed on AICS. WHMIS (Canada) CLASS D-2B: Material causing other toxic effects (TOXIC). Other Classifications R36- Irritating to eyes.S26- In case of contact with eyes, rinse DSCL (EEC) immediately with plenty of water and seek medical advice. Health Hazard HMIS (U.S.A.)2 National Fire Protection 1 Flammability 1 Association (U.S.A.) Fire Hazard 2 0 Reactivity Health Reactivity 0 Specific hazard Personal Protection E WHMIS (Canada) (Pictograms) DSCL (Europe) (Pictograms) TDG(Canada) (Pictograms) ADR (Europe) (Pictograms) Protective Equipment Gloves. Uric Acid, Sodium Salt Lab coat. Dust respirator. Be sure to use an approved/certified respirator or equivalent. Wear appropriate respirator when ventilation is inadequate. SECTION 16 - ADDITIONAL INFORMATION N/A |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | YU7062000 |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Molecular Mechanisms Underlying Urate-Induced Enhancement of Kv1.5 Channel Expression in HL-1 Atrial Myocytes.
Circ. J. 79 , 2659-68, (2015) Hyperuricemia induces endothelial dysfunction, oxidative stress and inflammation, increasing cardiovascular morbidities. It also raises the incidence of atrial fibrillation; however, underlying mechan... |
|
|
Direct radical scavenging activity of benzbromarone provides beneficial antioxidant properties for hyperuricemia treatment.
Biol. Pharm. Bull. 38(3) , 487-92, (2015) Uric acid exerts an important antioxidant effect against external oxidative stress under physiological conditions. However, uric acid itself can increase oxidative stress via reduced nicotinamide aden... |
| Harnsaeure,Natrium-Salz |
| uric acid,sodium-salt |
| 1H-Purine-2,6,8(3H)-trione, 7,9-dihydro-, sodium salt (1:1) |
| uric |
| SODIUM ACID URATE SODIUM SALT |
| SODIUM ACID URATE |
| MFCD00067310 |
| uricacidsodium |
| URIC ACID MONOSODIUM SALT |
| Uric acid, monosodium salt |
| Monosodium Urate |
| Sodium 6,8-dioxo-6,7,8,9-tetrahydro-3H-purin-2-olate |
| Uric acid monosodium |
| Sodium Urate |
| 3H-Purine-6,8-dione, 7,9-dihydro-2-hydroxy-, sodium salt (1:1) |