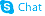Primidone
Primidone structure
|
Common Name | Primidone | ||
|---|---|---|---|---|
| CAS Number | 125-33-7 | Molecular Weight | 218.25200 | |
| Density | 1.138g/cm3 | Boiling Point | 520.7ºC at 760mmHg | |
| Molecular Formula | C12H14N2O2 | Melting Point | 281-282°C | |
| MSDS | Chinese USA | Flash Point | 228.2ºC | |
| Symbol |


GHS07, GHS08 |
Signal Word | Warning | |
Use of PrimidonePrimidone is an anticonvulsant of the pyrimidinedione class.Target: GABA ReceptorPrimidone is an anticonvulsant of the pyrimidinedione class, the active metabolites of which, phenobarbital (minor) and phenylethylmalonamide (PEMA) (major), are also anticonvulsants. It is believed to work via interactions with voltage-gated sodium channels which inhibit high-frequency repetitive firing of action potentials [1]. The effect of primidone in essential tremor is not mediated by PEMA.[76] The major metabolite, phenobarbital, is also a potent anticonvulsant in its own right and likely contributes to primidone's effects in many forms of epilepsy [2]. Primidone and the other enzyme-inducing anticonvulsants can cut the half-life of antipyrine roughly in half (6.2 ± 1.9 h vs. 11.2 ± 4.2 h), and increases the clearance rate by almost 70%. Phenobarbital reduces the half-life to 4.8 ± 1.3 and increases the clearance by almost 109% [3]. |
| Name | primidone |
|---|---|
| Synonym | More Synonyms |
| Description | Primidone is an anticonvulsant of the pyrimidinedione class.Target: GABA ReceptorPrimidone is an anticonvulsant of the pyrimidinedione class, the active metabolites of which, phenobarbital (minor) and phenylethylmalonamide (PEMA) (major), are also anticonvulsants. It is believed to work via interactions with voltage-gated sodium channels which inhibit high-frequency repetitive firing of action potentials [1]. The effect of primidone in essential tremor is not mediated by PEMA.[76] The major metabolite, phenobarbital, is also a potent anticonvulsant in its own right and likely contributes to primidone's effects in many forms of epilepsy [2]. Primidone and the other enzyme-inducing anticonvulsants can cut the half-life of antipyrine roughly in half (6.2 ± 1.9 h vs. 11.2 ± 4.2 h), and increases the clearance rate by almost 70%. Phenobarbital reduces the half-life to 4.8 ± 1.3 and increases the clearance by almost 109% [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.138g/cm3 |
|---|---|
| Boiling Point | 520.7ºC at 760mmHg |
| Melting Point | 281-282°C |
| Molecular Formula | C12H14N2O2 |
| Molecular Weight | 218.25200 |
| Flash Point | 228.2ºC |
| Exact Mass | 218.10600 |
| PSA | 58.20000 |
| LogP | 1.19550 |
| Vapour Pressure | 6.08E-11mmHg at 25°C |
| Index of Refraction | 1.528 |
| Storage condition | Store at RT |
| Water Solubility | <0.1 g/100 mL at 19 ºC |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H351 |
| Precautionary Statements | P280-P301 + P312 + P330 |
| Personal Protective Equipment | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S22-S36-S45 |
| RIDADR | 3249 |
| WGK Germany | 3 |
| RTECS | UV9100000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| Precursor 9 | |
|---|---|
| DownStream 3 | |
|
Optimized removal of dissolved organic carbon and trace organic contaminants during combined ozonation and artificial groundwater recharge.
Water Res. 46(18) , 6059-68, (2012) Pilot scale experiments using an 8 g/h ozonation unit and a 1.4 m(2) slow sand filter have demonstrated that the combination of ozonation and artificial groundwater recharge is suitable for efficient ... |
|
|
Carrier Mediated Distribution System (CAMDIS): a new approach for the measurement of octanol/water distribution coefficients.
Eur. J. Pharm. Sci. 68 , 68-77, (2015) Here we present a miniaturized assay, referred to as Carrier-Mediated Distribution System (CAMDIS) for fast and reliable measurement of octanol/water distribution coefficients, log D(oct). By introduc... |
|
|
How did phenobarbital's chemical structure affect the development of subsequent antiepileptic drugs (AEDs)?
Epilepsia 53 Suppl 8 , 3-11, (2012) Phenobarbital has been in clinical use as an antiepileptic drug (AED) since 1912. The initial clinical success of phenobarbital and other barbiturates affected the design of subsequent AEDs (e.g., phe... |
| Prysoline |
| Primidon |
| Mysoline |
| Misodine |
| MFCD00038662 |
| Sertan |
| Primidone |
| EINECS 204-737-0 |
| 5-ethyl-5-phenyl-dihydro-pyrimidine-4,6-dione |
| Mylepsinum |
| Liskantin |
| Mizodin |
| 5-ethyl-5-phenyl-1,3-diazinane-4,6-dione |
| Primaclone |
| 5-Ethyl-5-phenylhexahydropyrimidine-4,6-dione |
| 2-Deoxyphenobarbital |
| 2-Desoxyphenobarbital,5-Ethyl-5-phenylhexahydropyrimidine-4,6-dione |
 CAS#:2753-74-4
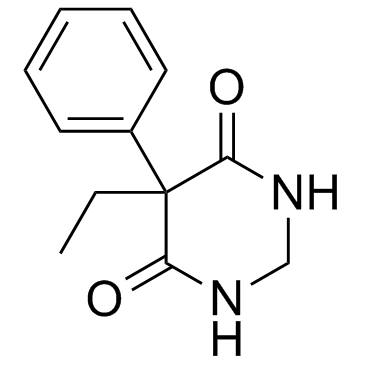
CAS#:2753-74-4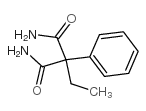 CAS#:7206-76-0
CAS#:7206-76-0 CAS#:77287-34-4
CAS#:77287-34-4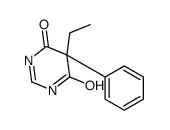 CAS#:694447-38-6
CAS#:694447-38-6 CAS#:857411-80-4
CAS#:857411-80-4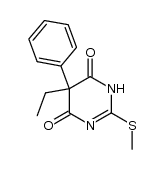 CAS#:343339-89-9
CAS#:343339-89-9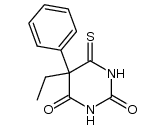 CAS#:60024-01-3
CAS#:60024-01-3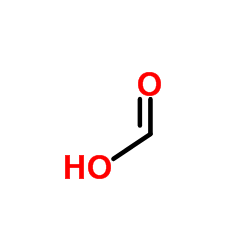 CAS#:64-18-6
CAS#:64-18-6 CAS#:50-06-6
CAS#:50-06-6 CAS#:75524-08-2
CAS#:75524-08-2