Protopine
Modify Date: 2025-08-21 12:15:40
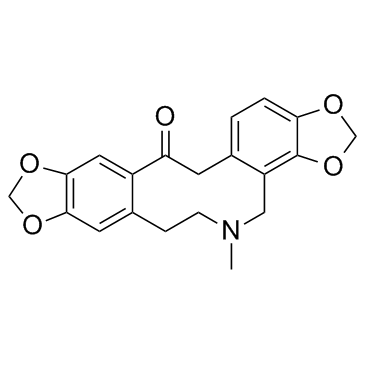
Protopine structure
|
Common Name | Protopine | ||
|---|---|---|---|---|
| CAS Number | 130-86-9 | Molecular Weight | 353.369 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 547.5±49.0 °C at 760 mmHg | |
| Molecular Formula | C20H19NO5 | Melting Point | 211ºC | |
| MSDS | Chinese USA | Flash Point | 284.9±29.8 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of ProtopineProtopine, an isoquinoline alkaloid contained in plants in northeast Asia.IC50 Value:Target:In vitro: Protopine was found to reduce nitric oxide (NO), cyclooxygenase-2 (COX-2), and prostaglandin E(2) (PGE(2)) production by LPS-stimulated Raw 264.7 cells, without a cytotoxic effect. Pre-treatment of Raw 264.7 cells with protopine reduced the production of pro-inflammatory cytokines [2]. Protopine is a novel microtubule stabilizer with anticancer activity in HRPC cells through apoptotic pathway by modulating Cdk1 activity and Bcl-2 family of proteins [3]. In HepG2 cells, protopine significantly increased CYP1A1 mRNA levels after 24h exposure at concentrations from 25 and 10 μM. Protopine also dose-dependently increased CYP1A1 and CYP1A2 mRNA levels in human hepatocytes [4].In vivo: Assays were performed on MDA-MB-231 human breast cancer cells, and the result showed that protopine exhibited anti-adhesive and anti-invasion effects in MDA-MB-231 cells; after treatment with protopine for 90 min, the expression of EGFR, ICAM-1, αv-integrin, β1-integrin and β5-integrin were remarkably reduced [1]. |
| Name | protopine |
|---|---|
| Synonym | More Synonyms |
| Description | Protopine, an isoquinoline alkaloid contained in plants in northeast Asia.IC50 Value:Target:In vitro: Protopine was found to reduce nitric oxide (NO), cyclooxygenase-2 (COX-2), and prostaglandin E(2) (PGE(2)) production by LPS-stimulated Raw 264.7 cells, without a cytotoxic effect. Pre-treatment of Raw 264.7 cells with protopine reduced the production of pro-inflammatory cytokines [2]. Protopine is a novel microtubule stabilizer with anticancer activity in HRPC cells through apoptotic pathway by modulating Cdk1 activity and Bcl-2 family of proteins [3]. In HepG2 cells, protopine significantly increased CYP1A1 mRNA levels after 24h exposure at concentrations from 25 and 10 μM. Protopine also dose-dependently increased CYP1A1 and CYP1A2 mRNA levels in human hepatocytes [4].In vivo: Assays were performed on MDA-MB-231 human breast cancer cells, and the result showed that protopine exhibited anti-adhesive and anti-invasion effects in MDA-MB-231 cells; after treatment with protopine for 90 min, the expression of EGFR, ICAM-1, αv-integrin, β1-integrin and β5-integrin were remarkably reduced [1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 547.5±49.0 °C at 760 mmHg |
| Melting Point | 211ºC |
| Molecular Formula | C20H19NO5 |
| Molecular Weight | 353.369 |
| Flash Point | 284.9±29.8 °C |
| Exact Mass | 353.126312 |
| PSA | 57.23000 |
| LogP | 3.76 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.613 |
| InChIKey | GPTFURBXHJWNHR-UHFFFAOYSA-N |
| SMILES | CN1CCc2cc3c(cc2C(=O)Cc2ccc4c(c2C1)OCO4)OCO3 |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 + P330 |
| Safety Phrases | 24/25 |
| RIDADR | UN 1544 |
| RTECS | VS2800000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| Precursor 4 | |
|---|---|
| DownStream 5 | |
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1100
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1101
|
|
Name: qHTS for inhibitors of Vif-A3G interactions: Validation
Source: NCGC
Target: DNA dC->dU-editing enzyme APOBEC-3G [Homo sapiens]
External Id: VIFG001
|
|
Name: Primary Screen Inhibitors of CD40 Signaling in BL2 Cells Measured in Cell-Based Syste...
Source: Broad Institute
Target: N/A
External Id: 7124-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
|
Name: High-throughput screen for inhibitors of the GIV GBA-motif interaction with Galpha-i ...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS1303
|
|
Name: qHTS for Inhibitors of binding or entry into cells for Marburg Virus
Source: NCGC
Target: gene 4 small orf - Marburg virus
External Id: VSVM-OFFLINE
|
|
Name: qHTS for Inhibitors of binding or entry into cells for Lassa Virus
Source: NCGC
Target: N/A
External Id: VSVL-OFFLINE
|
|
Name: Validation qHTS for agonist of cAMP-regulated guanine nucleotide exchange factor 3 (E...
Source: NCGC
External Id: epac1-activator-v
|
Total 28, Current Page 1 of 3
1
2
3
| 7-Methyl-6,8,9,16-tetrahydrobis[1,3]benzodioxolo[4,5-c:5',6'-g]azecin-15(7H)-one |
| Protopine |
| 7-Methyl-6,8,9,16-tetrahydro[1,3]dioxolo[5,6]benzo[1,2-c][1,3]dioxolo[4,5]benzo[1,2-g]azecin-15(7H)-on |
| Biflorine |
| Protopin |
| EINECS 204-999-6 |
| 4,6,7,14-Tetrahydro-5-methyl-bis[1,3]benzodioxolo[4,5-c:5',6'-g]azecin-13(5H)-one |
| BIFLORIN |
| Hypercorine |
| 4,6,7,14-Tetrahydro-5-Methyl |
| Tangeratin |
| Corydinine |
| Corydalis C |
 CAS#:64191-02-2
CAS#:64191-02-2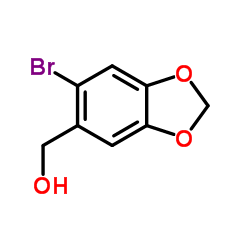 CAS#:6642-34-8
CAS#:6642-34-8![5-BROMO-6-(CHLOROMETHYL)BENZO[D][1,3]DIOXOLE Structure](https://image.chemsrc.com/caspic/226/64603-67-4.png) CAS#:64603-67-4
CAS#:64603-67-4 CAS#:41431-71-4
CAS#:41431-71-4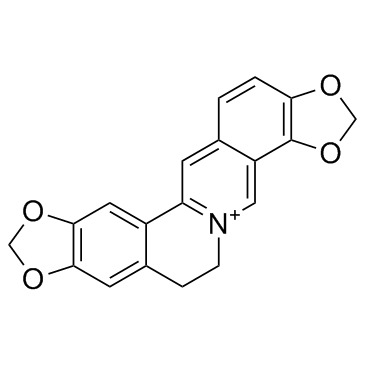 CAS#:3486-66-6
CAS#:3486-66-6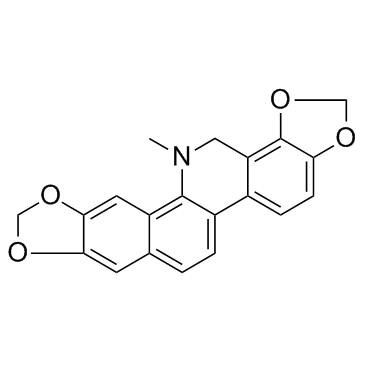 CAS#:3606-45-9
CAS#:3606-45-9 CAS#:2447-54-3
CAS#:2447-54-3 CAS#:6020-18-4
CAS#:6020-18-4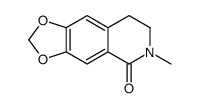 CAS#:552-29-4
CAS#:552-29-4
