Ziprasidone Hydrochloride Monohydrate
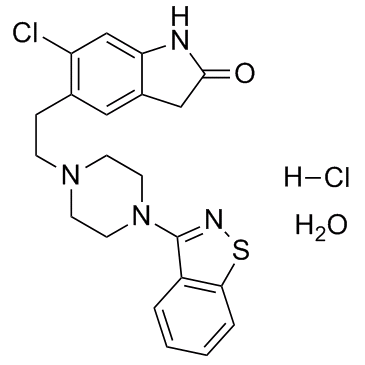
Ziprasidone Hydrochloride Monohydrate structure
|
Common Name | Ziprasidone Hydrochloride Monohydrate | ||
|---|---|---|---|---|
| CAS Number | 138982-67-9 | Molecular Weight | 467.412 | |
| Density | N/A | Boiling Point | 554.8ºC at 760 mmHg | |
| Molecular Formula | C21H24Cl2N4O2S | Melting Point | 300°C | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |


GHS07, GHS08 |
Signal Word | Warning | |
Use of Ziprasidone Hydrochloride MonohydrateZiprasidone(CP88059) is a combined 5-HT (serotonin) and dopamine receptor antagonist which exhibits potent effects of antipsychotic activity.Target: 5-HT receptor; Dopamine receptorZiprasidone (hydrochloride) is the salt form of ziprasidone, which possesses an in vitro 5-HT2A/dopamine D2 receptor affinity ratio higher than any clinically available antipsychotic agent. In vivo, ziprasidone antagonizes 5-HT2A receptor-induced head twitch with 6-fold higher potency than for blockade of d-amphetamine-induced hyperactivity, a measure of central dopamine D2 receptor antagonism. Ziprasidone also has high affinity for the 5-HT1A, 5-HT1D and 5-HT2C receptor subtypes, which may further enhance its therapeutic potential [1]. Ziprasidone sulfoxide and sulfone were the major metabolites in human serum. The affinities of the sulfoxide and sulfone metabolites for 5-HT2 and D2 receptors are low with respect to ziprasidone, and are thus unlikely to contribute to its antipsychotic effects [2]. Ziprasidone was associated with significant differential adverse effects relative to placebo in BPM, BPD, and schizophrenia with no significant difference in weight gain in all 3 groups. Self-reported somnolence was increased across the 3 conditions. Subjects with BPM were more vulnerable to EPS than those with BPD or schizophrenia [3].Clinical indications: Bipolar I disorder; Bipolar disorder; Mania; SchizophreniaFDA Approved Date: February 2001 |
| Name | ziprasidone hydrochloride hydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Ziprasidone(CP88059) is a combined 5-HT (serotonin) and dopamine receptor antagonist which exhibits potent effects of antipsychotic activity.Target: 5-HT receptor; Dopamine receptorZiprasidone (hydrochloride) is the salt form of ziprasidone, which possesses an in vitro 5-HT2A/dopamine D2 receptor affinity ratio higher than any clinically available antipsychotic agent. In vivo, ziprasidone antagonizes 5-HT2A receptor-induced head twitch with 6-fold higher potency than for blockade of d-amphetamine-induced hyperactivity, a measure of central dopamine D2 receptor antagonism. Ziprasidone also has high affinity for the 5-HT1A, 5-HT1D and 5-HT2C receptor subtypes, which may further enhance its therapeutic potential [1]. Ziprasidone sulfoxide and sulfone were the major metabolites in human serum. The affinities of the sulfoxide and sulfone metabolites for 5-HT2 and D2 receptors are low with respect to ziprasidone, and are thus unlikely to contribute to its antipsychotic effects [2]. Ziprasidone was associated with significant differential adverse effects relative to placebo in BPM, BPD, and schizophrenia with no significant difference in weight gain in all 3 groups. Self-reported somnolence was increased across the 3 conditions. Subjects with BPM were more vulnerable to EPS than those with BPD or schizophrenia [3].Clinical indications: Bipolar I disorder; Bipolar disorder; Mania; SchizophreniaFDA Approved Date: February 2001 |
|---|---|
| Related Catalog | |
| References |
[5]. Ziprasidone |
| Boiling Point | 554.8ºC at 760 mmHg |
|---|---|
| Melting Point | 300°C |
| Molecular Formula | C21H24Cl2N4O2S |
| Molecular Weight | 467.412 |
| Exact Mass | 466.099701 |
| PSA | 85.94000 |
| LogP | 4.68760 |
| Vapour Pressure | 2.38E-12mmHg at 25°C |
| InChIKey | ZCBZSCBNOOIHFP-UHFFFAOYSA-N |
| SMILES | Cl.O.O=C1Cc2cc(CCN3CCN(c4nsc5ccccc45)CC3)c(Cl)cc2N1 |
| Storage condition | Room temp |
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H317-H373 |
| Precautionary Statements | P260-P280 |
| Target Organs | Central nervous system, Liver |
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38:Irritating to eyes, respiratory system and skin . |
| Safety Phrases | S24/25 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2934999090 |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Gingival pain: an unusual side effect of ziprasidone.
BMJ Case Rep. 2013 , doi:10.1136/bcr-2012-007577, (2013) The patient is a 52-year-old man with schizophrenia who developed severe, unremitting gingival pain after his ziprasidone dosage was increased from 80 to 120 mg. His physical examination and laborator... |
|
|
A thorough QTc study of 3 doses of iloperidone including metabolic inhibition via CYP2D6 and/or CYP3A4 and a comparison to quetiapine and ziprasidone.
J. Clin. Psychopharmacol. 33(1) , 3-10, (2013) The potential for iloperidone, a D2/5-HT2A antipsychotic, to affect the heart rate-corrected QT interval (QTc) was assessed in the absence and presence of metabolic inhibitors in a randomized, open-la... |
|
|
Association between second-generation antipsychotics and changes in body mass index in adolescents.
J. Adolesc. Health 52(3) , 336-43, (2013) To assess the association of second-generation antipsychotics (SGAs) with changes in body mass index (BMI) among adolescents compared with a matched untreated comparison group.A retrospective cohort s... |
|
Name: qHTS to identify inhibitors of the New Delhi Metallo-beta-lactamase (NDM): assay vali...
Source: NCGC
External Id: adst_MBL_Abs_LOPAC_o1
|
|
Name: qHTS Assay for Inhibitors of the Phosphatase Activity of Eya2
Source: NCGC
Target: eyes absent homolog 2 isoform a [Homo sapiens]
External Id: EYA2477
|
|
Name: qHTS Assay for Inhibitors of Mammalian Selenoprotein Thioredoxin Reductase 1 (TrxR1)
Source: NCGC
Target: thioredoxin reductase [Rattus norvegicus]
External Id: TRXR193
|
|
Name: qHTS Assay for Substrates of Mammalian Selenoprotein Thioredoxin Reductase 1 (TrxR1)
Source: NCGC
Target: thioredoxin reductase [Rattus norvegicus]
External Id: TRXR188
|
|
Name: qHTS Validation Assay for Inhibitors of Ubiquitin-specific Protease USP2a Using CHOP2...
Source: NCGC
Target: ubiquitin carboxyl-terminal hydrolase 2 isoform a [Homo sapiens]
External Id: UBCH001
|
|
Name: qHTS assay for measurement of the activity of small molecule inhibitors on Zika virus...
Source: NCGC
Target: N/A
External Id: ZIK097
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
|
Name: Enzymatic assay of human HDAC6 with commercial peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808149
|
|
Name: Enzymatic assay of human HDAC6 with custom peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808150
|
| 5-(2-(4-(1,2-benzisothiazol-3-yl)piperazinyl)ethyl)-6-chlorooxindole Monohydrochloride Monohydrate |
| cp 88059-1 |
| 5-[2-[4-(1,2-Benzisothiazol-3yl)-1-piperazinyl]ethyl]-6-chloro-1,3-dihydro-2H-indol-2-one hydrochloride |
| Ziprasidone |
| Ziprasidone Hydrochloride Monohydrate |
| 5-[2-[4-(1,2-Benzisothiazol-3-yl)-1-piperazinyl]ethyl]-6-chloro-1,3-dihydro-2H-indol-2-on Monohydrochloride Monohydrate |
| 5-{2-[4-(1,2-Benzothiazol-3-yl)piperazin-1-yl]ethyl}-6-chloro-1,3-dihydro-2H-indol-2-one hydrochloride hydrate |
| 5-{2-[4-(1,2-Benzothiazol-3-yl)-1-piperazinyl]ethyl}-6-chloro-1,3-dihydro-2H-indol-2-one hydrochloride hydrate |
| Ziprasidone hydrochloride |
| MFCD06795476 |
| 2H-Indol-2-one, 5-[2-[4-(1,2-benzisothiazol-3-yl)-1-piperazinyl]ethyl]-6-chloro-1,3-dihydro-, hydrochloride, hydrate (1:1:1) |
| Ziprasidone (hydrochloride monohydrate) |