CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
RH6314000
-
CHEMICAL NAME :
-
D-erythro-D-Galacto-octopyranoside, methyl-6,8-dideoxy-6-(1-methyl-4-propyl-L-2- pyrrolidinecarboxamido)-1-thio-, trans-alpha-
-
CAS REGISTRY NUMBER :
-
154-21-2
-
LAST UPDATED :
-
199706
-
DATA ITEMS CITED :
-
9
-
MOLECULAR FORMULA :
-
C18-H34-N2-O6-S
-
MOLECULAR WEIGHT :
-
406.60
-
WISWESSER LINE NOTATION :
-
T6OTJ BS1 CQ DQ EQ FYYQ1&MV- BT5NTJ A1 D3
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
1 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
85ERAY "Antibiotics: Origin, Nature, and Properties," Korzyoski, T., et al., eds., Washington, DC, American Soc. for Microbiology, 1978 Volume(issue)/page/year: 1,186,1978
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
1900 mg/kg
-
TOXIC EFFECTS :
-
Brain and Coverings - recordings from specific areas of CNS Blood - other changes
-
REFERENCE :
-
ANKHEW Antibiotiki i Khimioterapiya. Antibiotics and Chemotherapy. (V/O Me zhdunarodnaya Kniga, 113095 Moskow, USSR) V.33- 1988- Volume(issue)/page/year: 35(2),40,1990
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
9778 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E. First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year: 18,185,1971
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
13900 mg/kg
-
TOXIC EFFECTS :
-
Brain and Coverings - recordings from specific areas of CNS Blood - other changes
-
REFERENCE :
-
ANKHEW Antibiotiki i Khimioterapiya. Antibiotics and Chemotherapy. (V/O Me zhdunarodnaya Kniga, 113095 Moskow, USSR) V.33- 1988- Volume(issue)/page/year: 35(2),40,1990
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
1 gm/kg
-
TOXIC EFFECTS :
-
Behavioral - convulsions or effect on seizure threshold
-
REFERENCE :
-
AACHAX Antimicrobial Agents and Chemotherapy (1961-70). (Ann Arbor, MI) 1961-70. For publisher information, see AMACCQ. Volume(issue)/page/year: -, 554,1962
-
TYPE OF TEST :
-
LDLo - Lowest published lethal dose
-
ROUTE OF EXPOSURE :
-
Intramuscular
-
SPECIES OBSERVED :
-
Rodent - rabbit
-
DOSE/DURATION :
-
200 ug/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
RMVEAG Recueil de Medecine Veterinaire. (Masson Pub. USA, Inc., Year Book Medical Pub., 35 E. Wacker Dr., Chicago IL 60601) V.1- 1824- Volume(issue)/page/year: 156,915,1980
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - guinea pig
-
DOSE/DURATION :
-
1800 mg/kg
-
TOXIC EFFECTS :
-
Brain and Coverings - recordings from specific areas of CNS Blood - other changes
-
REFERENCE :
-
ANKHEW Antibiotiki i Khimioterapiya. Antibiotics and Chemotherapy. (V/O Me zhdunarodnaya Kniga, 113095 Moskow, USSR) V.33- 1988- Volume(issue)/page/year: 35(2),40,1990 *** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA *** NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA : NOES - National Occupational Exposure Survey (1983) NOES Hazard Code - X3678 No. of Facilities: 147 (estimated) No. of Industries: 2 No. of Occupations: 4 No. of Employees: 1414 (estimated) No. of Female Employees: 161 (estimated)
|
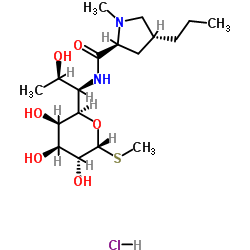
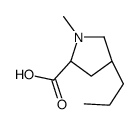 CAS#:13380-36-4
CAS#:13380-36-4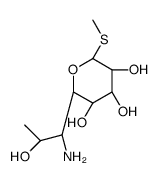 CAS#:14810-93-6
CAS#:14810-93-6 CAS#:13380-37-5
CAS#:13380-37-5