chloropyrimidine
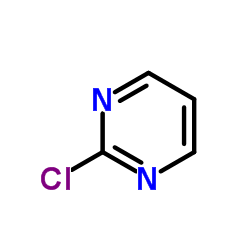
chloropyrimidine structure
|
Common Name | chloropyrimidine | ||
|---|---|---|---|---|
| CAS Number | 1722-12-9 | Molecular Weight | 114.533 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 199.2±0.0 °C at 760 mmHg | |
| Molecular Formula | C4H3ClN2 | Melting Point | 63-66 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 109.2±4.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 2-Chloropyrimidine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 199.2±0.0 °C at 760 mmHg |
| Melting Point | 63-66 °C(lit.) |
| Molecular Formula | C4H3ClN2 |
| Molecular Weight | 114.533 |
| Flash Point | 109.2±4.3 °C |
| Exact Mass | 113.998474 |
| PSA | 25.78000 |
| LogP | 0.36 |
| Vapour Pressure | 0.5±0.3 mmHg at 25°C |
| Index of Refraction | 1.535 |
| InChIKey | UNCQVRBWJWWJBF-UHFFFAOYSA-N |
| SMILES | Clc1ncccn1 |
| Storage condition | Refrigerator |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H319 |
| Precautionary Statements | P301 + P312 + P330-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/38 |
| Safety Phrases | S26-S37/39 |
| RIDADR | 2811 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2933599090 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933599090 |
|---|---|
| Summary | 2933599090. other compounds containing a pyrimidine ring (whether or not hydrogenated) or piperazine ring in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Cobalt-catalyzed cross-coupling between in situ prepared arylzinc halides and 2-chloropyrimidine or 2-chloropyrazine.
J. Org. Chem. 74(8) , 3221-4, (2009) A cobalt-catalyzed cross-coupling of aryl halides with 2-chloropyrimidines or 2-chloropyrazines is reported in satisfactory to high yields. The key step of this procedure is the formation of aromatic ... |
|
|
Ru-catalysed C-H arylation of indoles and pyrroles with boronic acids: scope and mechanistic studies.
Chemistry 21(14) , 5380-6, (2015) The Ru-catalysed C2-H arylation of indoles and pyrroles by using boronic acids under oxidative conditions is reported. This reaction can be applied to tryptophan derivatives and tolerates a wide range... |
|
|
Expedient parallel synthesis of 2-amino-4-heteroarylpyrimidines.
Org. Lett. 7(19) , 4113-6, (2005) [reaction: see text] An expedient synthesis of diverse 2-amino-4-heteroarylpyrimidines via a 2-chloropyrimidine intermediate is described. A series of potentially biologically active analogues have be... |
| 2-Chloro-4-deoxyuracil |
| 2-Chloro-pyrimindine |
| EINECS 217-020-2 |
| chloro-pyrimidine |
| 2-Chloropyrimindine |
| MFCD00006060 |
| chloropyrimidine |
| 2-pyrimidyl chloride |
| 2-Chloropyrimidine |
| 2-Cl-pyrimidine |
| Pyrimidine, 2-chloro- |
| 2CPY X HCL |
| 2-Chloro |
| pyrimidin-2-yl chloride |
| 2-chloro-pyrimidine |
| 2-chloro-pyrimidin |
| 2-choropyrimidine |
| PYRIMIDINE,2-CHLORO |
| 2-pyrimidinyl chloride |
| 2-chloropyrimide |
 CAS#:56-05-3
CAS#:56-05-3 CAS#:55-81-2
CAS#:55-81-2 CAS#:702-23-8
CAS#:702-23-8 CAS#:18217-00-0
CAS#:18217-00-0 CAS#:1003845-06-4
CAS#:1003845-06-4 CAS#:109-12-6
CAS#:109-12-6 CAS#:557-01-7
CAS#:557-01-7 CAS#:3934-20-1
CAS#:3934-20-1 CAS#:97-93-8
CAS#:97-93-8 CAS#:1088994-20-0
CAS#:1088994-20-0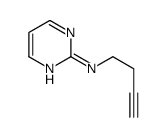 CAS#:111097-54-2
CAS#:111097-54-2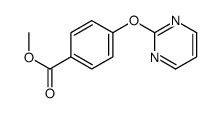 CAS#:1090587-89-5
CAS#:1090587-89-5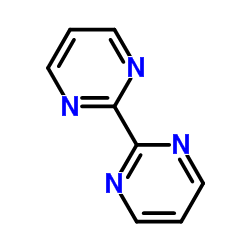 CAS#:34671-83-5
CAS#:34671-83-5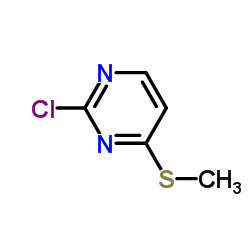 CAS#:49844-93-1
CAS#:49844-93-1 CAS#:145208-86-2
CAS#:145208-86-2 CAS#:151390-02-2
CAS#:151390-02-2 CAS#:5621-02-3
CAS#:5621-02-3 CAS#:141193-13-7
CAS#:141193-13-7 CAS#:151390-00-0
CAS#:151390-00-0
