5-Bromo-2-furaldehyde
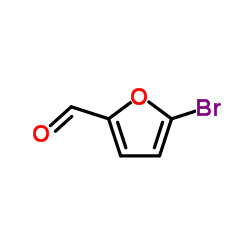
5-Bromo-2-furaldehyde structure
|
Common Name | 5-Bromo-2-furaldehyde | ||
|---|---|---|---|---|
| CAS Number | 1899-24-7 | Molecular Weight | 174.980 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 222.9±20.0 °C at 760 mmHg | |
| Molecular Formula | C5H3BrO2 | Melting Point | 82-85 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 88.6±21.8 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 5-bromofuran-2-carbaldehyde |
|---|---|
| Synonym | More Synonyms |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 222.9±20.0 °C at 760 mmHg |
| Melting Point | 82-85 °C(lit.) |
| Molecular Formula | C5H3BrO2 |
| Molecular Weight | 174.980 |
| Flash Point | 88.6±21.8 °C |
| Exact Mass | 173.931641 |
| PSA | 30.21000 |
| LogP | 1.77 |
| Vapour Pressure | 0.1±0.4 mmHg at 25°C |
| Index of Refraction | 1.573 |
| InChIKey | WJTFHWXMITZNHS-UHFFFAOYSA-N |
| SMILES | O=Cc1ccc(Br)o1 |
| Storage condition | Refrigerator |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H312-H315-H319-H332-H335 |
| Precautionary Statements | P261-P280-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R20/21/22;R36/37/38 |
| Safety Phrases | S26-S36-S36/37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2932190090 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2932190090 |
|---|---|
| Summary | 2932190090 other compounds containing an unfused furan ring (whether or not hydrogenated) in the structure VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:20.0% |
|
5-Substituted-2-furaldehydes: a synthetic protocol utilizing an organozinc route.
J. Org. Chem. 78(5) , 1984-93, (2013) Facile synthetic routes for the preparation of a wide range of 5-substituted 2-furaldehydes have been revealed. They were accomplished through either Pd-catalyzed cross-coupling reaction of various ar... |
|
|
From furans to anilines: toward one-pot two-step amination/diels-alder sequences.
J. Org. Chem. 73(6) , 2191-7, (2008) Selective metal-free amination and Diels-Alder reactions are described in the furan series, leading to polysubstituted anilines or to stable oxabicyclic adducts in high yield. Interestingly, anilines ... |
|
|
Efficient coupling of heteroaryl bromides with arylboronic acids in the presence of a palladium-tetraphosphine catalyst. Feuerstein M, et al.
Tetrahedron Lett. 42(23) , 5659-62, (2001)
|
| 5-Bromofurfural |
| 5-bromofuran-2-carbaldehyde |
| 5-Bromofurancarboxaldehyde |
| 5-bromo-2-furancarboxaldehyde |
| 5-bromofuran-2-yl carbaldehyde |
| 2-Furaldehyde, 5-bromo- |
| 2-Furancarboxaldehyde, 5-bromo- |
| 5'-bromo-furan-2'-carboxaldehyde |
| 5-Bromo-2-furaldehyde |
| 5-bromo-furfurol |
| 5-bromo-2-furfural |
| 2-Furancarboxaldehyde,5-bromo |
| MFCD00159501 |
 CAS#:179055-22-2
CAS#:179055-22-2 CAS#:4915-06-4
CAS#:4915-06-4 CAS#:35950-55-1
CAS#:35950-55-1 CAS#:98-01-1
CAS#:98-01-1 CAS#:1137088-24-4
CAS#:1137088-24-4 CAS#:613-75-2
CAS#:613-75-2 CAS#:585-70-6
CAS#:585-70-6![bis-(5-formyl-[2]furyl)-mercury Structure](https://image.chemsrc.com/caspic/312/854819-96-8.png) CAS#:854819-96-8
CAS#:854819-96-8![bis-(5-hydroxymethyl-[2]furyl)-mercury Structure](https://image.chemsrc.com/caspic/171/854753-06-3.png) CAS#:854753-06-3
CAS#:854753-06-3 CAS#:53355-29-6
CAS#:53355-29-6 CAS#:34035-03-5
CAS#:34035-03-5 CAS#:3857-25-8
CAS#:3857-25-8![5-[2-(TRIFLUOROMETHOXY)PHENYL]-2-FURALDEHYDE structure](https://image.chemsrc.com/caspic/470/306936-00-5.png) CAS#:306936-00-5
CAS#:306936-00-5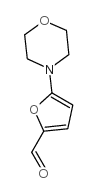 CAS#:3680-96-4
CAS#:3680-96-4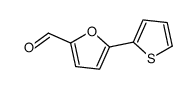 CAS#:32364-30-0
CAS#:32364-30-0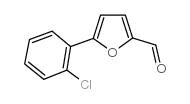 CAS#:34035-04-6
CAS#:34035-04-6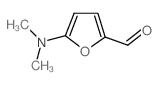 CAS#:3680-93-1
CAS#:3680-93-1 CAS#:82386-85-4
CAS#:82386-85-4 CAS#:436088-58-3
CAS#:436088-58-3
