5-Bromonicotinic acid
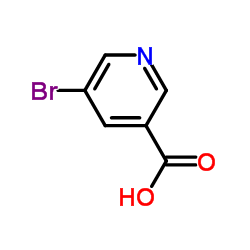
5-Bromonicotinic acid structure
|
Common Name | 5-Bromonicotinic acid | ||
|---|---|---|---|---|
| CAS Number | 20826-04-4 | Molecular Weight | 202.01 | |
| Density | 1.8±0.1 g/cm3 | Boiling Point | 328.5±27.0 °C at 760 mmHg | |
| Molecular Formula | C6H4BrNO2 | Melting Point | 178-180 °C(lit.) | |
| MSDS | USA | Flash Point | 152.5±23.7 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of 5-Bromonicotinic acid5-Bromonicotinic acid is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | 5-bromopyridine-3-carboxylic acid |
|---|---|
| Synonym | More Synonyms |
| Description | 5-Bromonicotinic acid is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Density | 1.8±0.1 g/cm3 |
|---|---|
| Boiling Point | 328.5±27.0 °C at 760 mmHg |
| Melting Point | 178-180 °C(lit.) |
| Molecular Formula | C6H4BrNO2 |
| Molecular Weight | 202.01 |
| Flash Point | 152.5±23.7 °C |
| Exact Mass | 200.942535 |
| PSA | 50.19000 |
| LogP | 1.22 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.617 |
| InChIKey | FQIUCPGDKPXSLL-UHFFFAOYSA-N |
| SMILES | O=C(O)c1cncc(Br)c1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | QT0868000 |
| HS Code | 2933399090 |
|
~96% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: WO2004/35545 A2, ; Page 45-46 ; |
|
~11% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Journal of Pharmaceutical Sciences, , vol. 84, # 9 p. 1120 - 1125 |
|
~89% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Tetrahedron: Asymmetry, , vol. 3, # 7 p. 871 - 901 |
|
~% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Journal of the American Chemical Society, , vol. 72, p. 393 |
|
~% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Journal of the American Chemical Society, , vol. 70, p. 2381,2383 |
|
~% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Journal of Organic Chemistry, , vol. 16, p. 1485,1491 |
|
~% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Synthesis, , # 4 p. 551 - 554 |
|
~% 
5-Bromonicotini... CAS#:20826-04-4 |
| Literature: Yakugaku Zasshi, , vol. 51, p. 542,571; dtsch. Ref. S. 73, 76 Chem.Abstr., , p. 5427 Journal fuer Praktische Chemie (Leipzig), , vol. <2> 138, p. 244,257 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Agonist lead identification for the high affinity niacin receptor GPR109a.
Bioorg. Med. Chem. Lett. 17 , 4914-9, (2007) A strategy for lead identification of new agonists of GPR109a, starting from known compounds shown to activate the receptor, is described. Early compound triage led to the formulation of a binding hyp... |
|
|
Iodopyridine-for-iodobenzene substitution for use with low molecular weight radiopharmaceuticals: application to m-iodobenzylguanidine.
Bioconjug. Chem. 9(6) , 758-64, (1998) Substituting a pyridine ring for a benzene ring in the acylation agent N-succinimidyl 3-iodobenzoate has resulted in a useful approach for the radiohalogenation of monoclonal antibodies, peptides, and... |
|
|
Drug-protein conjugates: haptenation of 1-methyl-10 alpha-methoxydihydrolysergol and 5-bromonicotinic acid to albumin for the production of epitope-specific monoclonal antibodies against nicergoline.
J. Pharm. Sci. 84(9) , 1120-5, (1995) Two types of monoclonal antibodies were used for the determination of nicergoline in biological matrices. The antibodies were prepared with the hydrolysis products 5-bromonicotinic acid and 1-methyl-1... |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Antibacterial activity against Klebsiella pneumoniae MDR ATCC 70063 (CO-ADD:GN_003); ...
Source: ChEMBL
Target: Klebsiella pneumoniae
External Id: CHEMBL4296186
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
| Nicotinic acid,5-bromo |
| 5-Bromonicotinic acid |
| 5-Bromo-3-pyridinecarboxylic Acid |
| 5-Bromopyridine-3-carboxylic acid,5-Bromonicotinic acid |
| 3-Pyridinecarboxylic acid,5-bromo |
| 3-bromo-5-pyridine-carboxylic acid |
| 5-bromonictotinic acid |
| 3-Bromo pyridine-5-carboxylic acid |
| 5-bromonicotininc acid |
| 5-Bromopyridine-3-carboxylic acid |
| EINECS 244-065-5 |
| MFCD00009783 |
| 5-Bromo Nicotinic Acid |
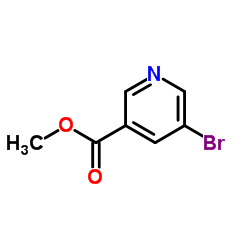
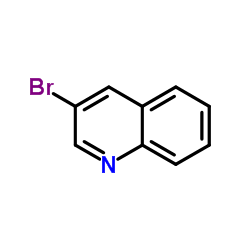
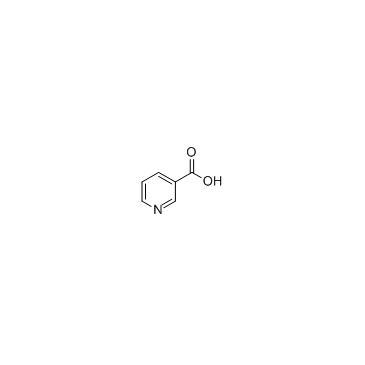
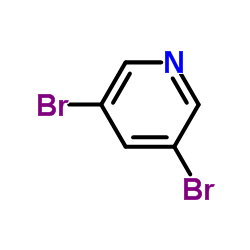

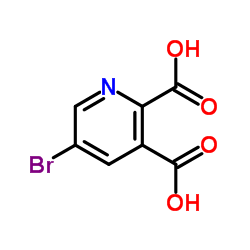
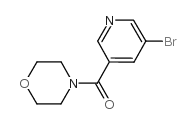 CAS#:342013-81-4
CAS#:342013-81-4 CAS#:342013-78-9
CAS#:342013-78-9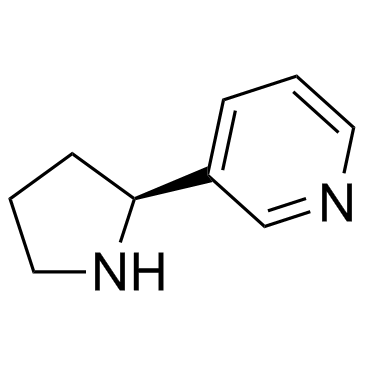 CAS#:494-97-3
CAS#:494-97-3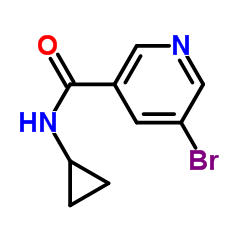 CAS#:385382-48-9
CAS#:385382-48-9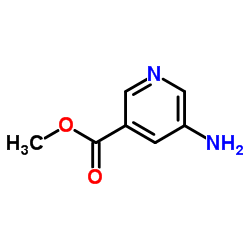 CAS#:36052-25-2
CAS#:36052-25-2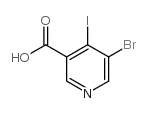 CAS#:491588-98-8
CAS#:491588-98-8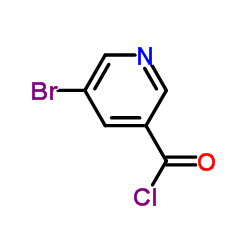 CAS#:39620-02-5
CAS#:39620-02-5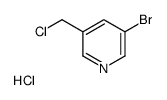 CAS#:39741-46-3
CAS#:39741-46-3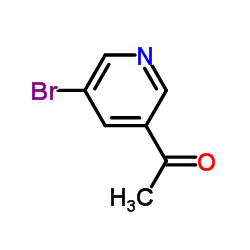 CAS#:38940-62-4
CAS#:38940-62-4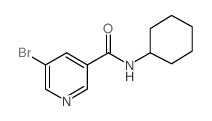 CAS#:342013-85-8
CAS#:342013-85-8
