CB 1954
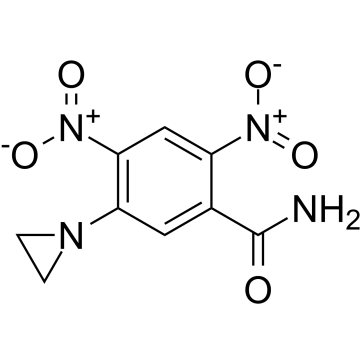
CB 1954 structure
|
Common Name | CB 1954 | ||
|---|---|---|---|---|
| CAS Number | 21919-05-1 | Molecular Weight | 252.184 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 427.2±45.0 °C at 760 mmHg | |
| Molecular Formula | C9H8N4O5 | Melting Point | 173 °C | |
| MSDS | USA | Flash Point | 212.2±28.7 °C | |
Use of CB 1954Tretazicar (CB 1954), an antitumor prodrug, is highly selective against the Walker 256 rat tumour line. Tretazicar is enzymatically activated to generate a bifunctional agent, which can form DNA-DNA interstrand cross-links. Tretazicar in rat cells involves the reduction of its 4-nitro group to a 4-hydroxylamine by the enzyme NAD(P)H:quinone oxidoreductase 1 (NQO1)[1][2]. |
| Name | 5-(aziridin-1-yl)-2,4-dinitrobenzamide |
|---|---|
| Synonym | More Synonyms |
| Description | Tretazicar (CB 1954), an antitumor prodrug, is highly selective against the Walker 256 rat tumour line. Tretazicar is enzymatically activated to generate a bifunctional agent, which can form DNA-DNA interstrand cross-links. Tretazicar in rat cells involves the reduction of its 4-nitro group to a 4-hydroxylamine by the enzyme NAD(P)H:quinone oxidoreductase 1 (NQO1)[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Tretazicar (CB 1954) (0.1-1000 μM; 3 days) has sensitivity for retrovirally transduced AB22 (AB22-nr) cells with an IC50 of 3 μM[3]. DNA cross-link formation in affected cells is a result of the bioactivation of the drug by the enzyme DT diaphorase (NAD(P)H dehydro-genase (quinone)) in the Walker cells which reduces the 4-nitro group of Tretazicar. The product of this reaction is a difunctional alkylating agent, 5-aziridin-1-yl-4-hydroxylamino-2-nitrobenzamide[4]. |
| In Vivo | Tretazicar (CB 1954) (80 mg/kg; i.p. on days 2 and 9) results in a significant increase in survival[3]. Animal Model: Female BALB/c mice (AB22-nr, SKOV3 human ovarian tumour xenograft)[3] Dosage: 80 mg/kg Administration: i.p. on days 2 and 9 Result: The median survival of the AB22-nr was 49 days. Resulted in a significant increase in survival. |
| References |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 427.2±45.0 °C at 760 mmHg |
| Melting Point | 173 °C |
| Molecular Formula | C9H8N4O5 |
| Molecular Weight | 252.184 |
| Flash Point | 212.2±28.7 °C |
| Exact Mass | 252.049469 |
| PSA | 137.74000 |
| LogP | 1.28 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.715 |
| InChIKey | WOCXQMCIOTUMJV-UHFFFAOYSA-N |
| SMILES | NC(=O)c1cc(N2CC2)c([N+](=O)[O-])cc1[N+](=O)[O-] |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
An unusually cold active nitroreductase for prodrug activations
Bioorg. Med. Chem. 20(11) , 3540-50, (2012) A set of PCR primers based on the genome sequence were used to clone a gene encoding a hypothetical nitroreductases (named as Ssap-NtrB) from uropathogenic staphylococcus, Staphylococcus saprophyticus... |
|
|
uvrB gene deletion enhances SOS chromotest sensitivity for nitroreductases that preferentially generate the 4-hydroxylamine metabolite of the anti-cancer prodrug CB1954.
J. Biotechnol. 150(1) , 190-4, (2010) CB1954 is an anti-cancer prodrug that can be reduced at either of two nitro groups to form cytotoxic metabolites. We describe here two efficient and previously uncharacterized nitroreductases, YfkO fr... |
|
|
CB 1954: from the Walker tumor to NQO2 and VDEPT.
Curr. Pharm. Des. 9(26) , 2091-104, (2003) CB 1954 [5-(aziridin-1-yl)-2,4-dinitrobenzamide] has been the subject of continued interest for over 30 years. As an anti-cancer agent, it represents one of the very few examples of a compound that sh... |
|
Name: Partition coefficient at pH 7.4 by shake-flask method
Source: ChEMBL
Target: N/A
External Id: CHEMBL913785
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Antiproliferative activity against chinese hamster wild-type V79 NTR-ve cells after 1...
Source: ChEMBL
Target: V79
External Id: CHEMBL913786
|
|
Name: Antiproliferative activity against chinese hamster V79 NTRpuro cells after 18 hrs
Source: ChEMBL
Target: V79
External Id: CHEMBL913787
|
|
Name: Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL1266185
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Selectivity ratio of IC50 for V79 NTR-ve cells to IC50 for V79 NTRpuro cells
Source: ChEMBL
Target: V79
External Id: CHEMBL913788
|
|
Name: Ratio of Kcat to that of Km was determined
Source: ChEMBL
Target: N/A
External Id: CHEMBL627986
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Antiproliferative activity against human WiDr NTRneo cells after 18 hrs
Source: ChEMBL
Target: WiDr
External Id: CHEMBL913793
|
| 5-(1-Aziridinyl)-2,4-dinitrobenzamide |
| 2,4-Dinitro-5-ethyleneiminobenzamide |
| 2,4-Dinitroethyleneiminobenzamide |
| 5-(aziridyn-1-yl)-2,4-dinitrobenzamide |
| 5-Aziridinyl-2,4-dinitrobenzamide |
| CB-1954 |
| 5-(Aziridin-1-yl)-2,4-dinitrobenzamide |
| Tretazicar |
| 1 (5 carbamoyl 2,4 dinitrophenyl)aziridine |
| UNII-7865D5D01M |
| 5-aziridino-2,4-dinitrobenzamide |