H-Leu-OtBu.HCl
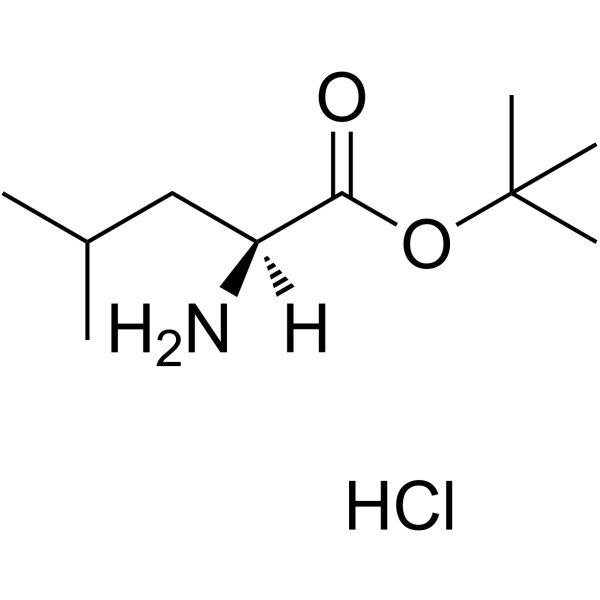
H-Leu-OtBu.HCl structure
|
Common Name | H-Leu-OtBu.HCl | ||
|---|---|---|---|---|
| CAS Number | 2748-02-9 | Molecular Weight | 223.740 | |
| Density | 0.929g/cm3 | Boiling Point | 222.4ºC at 760 mmHg | |
| Molecular Formula | C10H22ClNO2 | Melting Point | 167 °C | |
| MSDS | Chinese USA | Flash Point | 90.3ºC | |
Use of H-Leu-OtBu.HClH-Leu-OtBu.HCl is a leucine derivative[1]. |
| Name | L-Leucine tert-butyl ester hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | H-Leu-OtBu.HCl is a leucine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 0.929g/cm3 |
|---|---|
| Boiling Point | 222.4ºC at 760 mmHg |
| Melting Point | 167 °C |
| Molecular Formula | C10H22ClNO2 |
| Molecular Weight | 223.740 |
| Flash Point | 90.3ºC |
| Exact Mass | 223.133911 |
| PSA | 52.32000 |
| LogP | 3.20380 |
| Vapour Pressure | 0.102mmHg at 25°C |
| Index of Refraction | 1.444 |
| Storage condition | 2-8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2922499990 |
| HS Code | 2922499990 |
|---|---|
| Summary | HS:2922499990 other amino-acids, other than those containing more than one kind of oxygen function, and their esters; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
|
Glutamate formation via the leucine-to-glutamate pathway of rat pancreas.
Am. J. Physiol. Gastrointest. Liver Physiol. 306(11) , G938-46, (2014) The leucine-to-glutamate (Leu→Glu) pathway, which metabolizes the carbon atoms of l-leucine to form l-glutamate, was studied by incubation of rat tissue segments with l-[U-(14)C]leucine and estimation... |
|
|
Different EDC/NHS activation mechanisms between PAA and PMAA brushes and the following amidation reactions.
Langmuir 27 , 12058-12068, (2011) Infrared spectroscopy was applied to investigate the well-known EDC/NHS (N-ethyl-N'-(3-(dimethylamino)propyl)carbodiimide/N-hydroxysuccinimide) activation details of poly(acrylic acid) (PAA) and poly(... |
|
|
Reversal of stereoselectivity in the Cu-catalyzed conjugate addition reaction of dialkylzinc to cyclic enone in the presence of a chiral azolium compound.
J. Org. Chem. 75 , 5707-5715, (2010) Reversal of enantioselectivity in a Cu-catalyzed asymmetric conjugate addition reaction of dialkylzinc to cyclic enone with use of the same chiral ligand was successfully achieved. The reaction of 2-c... |
| L-Leucine t-Butyl Ether Hcl Salt |
| L-leucine tBu ester hydrochloride |
| tert-butyl L-leucinate hydrochloride |
| L-LEUCINET-BUTYL ESTER HYDROCHLORIDE |
| H-LEU-OTBU-OH |
| DL-leucine t-butyl ester hydrochloride |
| H-LEU-OTBU HYDROCHLORIDE |
| L-Leucine tert-Butyl Ester Hydrochloride |
| H-Leu-O-t-Bu hydrochloride |
| Leu-OBut*HCl |
| L-LEUCINE TERT-BUTYL ESTER HCL |
| LEUCINE-OTBU HCL |
| L-Leucine, 1,1-dimethylethyl ester, hydrochloride (1:1) |
| MFCD00038906 |
| H-Leu-OtBu HCl |
| L-LEU-O-TERT-BU HCL |
| H-Leu-OtBu,HCl |
| L-LEUCINE T-BUTYL ESTER |
| H-LEU-OBUT HCL |
| H-Leu-OtBu.HCl |
| L-LEUCINE-TERT-BUTYLESTER HYDROCHLORIDE |
| H-Leu-OtBu · HCl |
| L-LEUCINE T-BUTYL ESTER HYDROCHLORIDE |
| L-LEUCINE,1,1-DIMETHYLETHYL ESTER, HYDROCHLORIDE (1:1) |
| 2-Methyl-2-propanyl L-leucinate hydrochloride (1:1) |
| H-Leu-OtBu·HCl |
| L-Leucinetert-butyl ester hydrochloride |
| H-Leu-OtBu |
| H-L-Leu-OtBu*HCl |
| LeuOtBu.Hcl |
| Leu-Otbu.Hcl |
| H-LEU-OTBU . HCL |
| H-LEU-OBUT·HCL |
| L-Leucine tert·butyl ester hydrochloride |
| HCl*leucine tert butyl ester |
| L-LEUCINE T-BUTYL ESTER HCL |
| tert-Butyl L-leucinate hydrochloride (1:1) |
| L-LEUCINETERT-BTUYLESTERHCL |
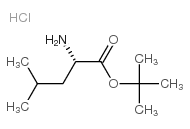 CAS#:21691-53-2
CAS#:21691-53-2 CAS#:58822-25-6
CAS#:58822-25-6 CAS#:81678-16-2
CAS#:81678-16-2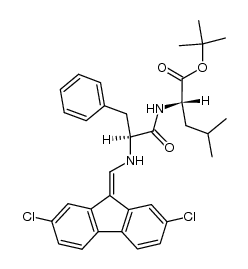 CAS#:122237-16-5
CAS#:122237-16-5
