α-Hydroxyglutaric acid
Modify Date: 2025-08-26 23:19:34
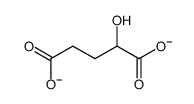
α-Hydroxyglutaric acid structure
|
Common Name | α-Hydroxyglutaric acid | ||
|---|---|---|---|---|
| CAS Number | 2889-31-8 | Molecular Weight | 146.09800 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C5H6O5-- | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of α-Hydroxyglutaric acidα-Hydroxyglutaric acid (2-Hydroxyglutarate) is an α-hydroxy acid form of glutaric acid. α-Hydroxyglutaric acid is a competitive inhibitor of multiple α-ketoglutarate-dependent dioxygenases, including histone demethylases and the TET family of 5-methlycytosine (5mC) hydroxylases[1]. |
| Name | 2-hydroxyglutaric acid |
|---|---|
| Synonym | More Synonyms |
| Description | α-Hydroxyglutaric acid (2-Hydroxyglutarate) is an α-hydroxy acid form of glutaric acid. α-Hydroxyglutaric acid is a competitive inhibitor of multiple α-ketoglutarate-dependent dioxygenases, including histone demethylases and the TET family of 5-methlycytosine (5mC) hydroxylases[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Isocitrate Dehydrogenase 1 (IDH1) and IDH2 mutations occur frequently in gliomas and acute myeloid leukemia, leading to simultaneous loss and gain of activities in the production of α-ketoglutarate (α-KG) and α-Hydroxyglutaric acid (2-Hydroxyglutarate), respectively[1]. α-Hydroxyglutaric acid (2-Hydroxyglutarate) inhibits the activity of multiple histone demethylases. α-Hydroxyglutaric acid occupies the same space as α-KG does in the active site of histone demethylases. α-Hydroxyglutaric acid (2-Hydroxyglutarate) inhibits the activity of TET 5-methylcytosine hydroxylases[1]. Treatment of U-87MG cells with α-Hydroxyglutaric acid (2-Hydroxyglutarate; 10-50 mM) increases HIF-1α and decreases endostatin[1]. |
| References |
| Molecular Formula | C5H6O5-- |
|---|---|
| Molecular Weight | 146.09800 |
| Exact Mass | 146.02200 |
| PSA | 100.49000 |
| InChIKey | HWXBTNAVRSUOJR-UHFFFAOYSA-N |
| SMILES | O=C(O)CCC(O)C(=O)O |
| 2-hydroxyglutarate |
 CAS#:110-94-1
CAS#:110-94-1 CAS#:1724-02-3
CAS#:1724-02-3![2-[3-(3H-1,3-benzothiazol-2-ylidene)prop-1-enyl]-1,3-benzothiazole structure](https://image.chemsrc.com/caspic/005/27698-70-0.png) CAS#:27698-70-0
CAS#:27698-70-0 CAS#:69134-53-8
CAS#:69134-53-8 CAS#:4344-84-7
CAS#:4344-84-7