Desmesterol
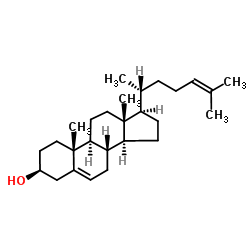
Desmesterol structure
|
Common Name | Desmesterol | ||
|---|---|---|---|---|
| CAS Number | 313-04-2 | Molecular Weight | 384.638 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 483.2±14.0 °C at 760 mmHg | |
| Molecular Formula | C27H44O | Melting Point | 121.5ºC | |
| MSDS | Chinese USA | Flash Point | 209.2±12.4 °C | |
Use of DesmesterolDesmosterol is a molecule similar to cholesterol. Desmosterol is the immediate precursor of cholesterol in the Bloch pathway of cholesterol biosynthesis. Desmosterol, as an endogenous metabolite, used to study cholesterol metabolism[1]. |
| Name | desmosterol |
|---|---|
| Synonym | More Synonyms |
| Description | Desmosterol is a molecule similar to cholesterol. Desmosterol is the immediate precursor of cholesterol in the Bloch pathway of cholesterol biosynthesis. Desmosterol, as an endogenous metabolite, used to study cholesterol metabolism[1]. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
[1]. Clark RM, et al. Desmosterol in human milk. Lipids. 1983;18(3):264-266. |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 483.2±14.0 °C at 760 mmHg |
| Melting Point | 121.5ºC |
| Molecular Formula | C27H44O |
| Molecular Weight | 384.638 |
| Flash Point | 209.2±12.4 °C |
| Exact Mass | 384.339203 |
| PSA | 20.23000 |
| LogP | 9.54 |
| Vapour Pressure | 0.0±2.8 mmHg at 25°C |
| Index of Refraction | 1.536 |
| InChIKey | AVSXSVCZWQODGV-DPAQBDIFSA-N |
| SMILES | CC(C)=CCCC(C)C1CCC2C3CC=C4CC(O)CCC4(C)C3CCC12C |
| Storage condition | -20°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| Precursor 10 | |
|---|---|
| DownStream 4 | |
|
A liquid chromatography-tandem mass spectrometry-based method for the simultaneous determination of hydroxy sterols and bile acids.
J. Chromatogr. A. 1371 , 184-95, (2014) Recently, hydroxy sterols and bile acids have gained growing interest as they are important regulators of energy homoeostasis and inflammation. The high number of different hydroxy sterols and bile ac... |
|
|
Sterol Composition in Infant Formulas and Estimated Intake.
J. Agric. Food Chem. 63 , 7245-51, (2015) Sterol contents in infant formulas (IFs) from the European market were determined, and their intakes by infants between 0 and 6 months were evaluated. Total animal sterols (mg/100 mL) ranged from 1.71... |
|
|
Analysis of hedgehog signaling in cerebellar granule cell precursors in a conditional Nsdhl allele demonstrates an essential role for cholesterol in postnatal CNS development.
Hum. Mol. Genet. 24 , 2808-25, (2015) NSDHL is a 3β-hydroxysterol dehydrogenase that is involved in the removal of two C-4 methyl groups in one of the later steps of cholesterol biosynthesis. Mutations in the gene encoding the enzyme are ... |
|
Name: Cytotoxicity against human A549 cells by MTT assay
Source: ChEMBL
Target: A549
External Id: CHEMBL1025019
|
|
Name: Cytotoxicity against human HT29 cells by MTT assay
Source: ChEMBL
Target: HT-29
External Id: CHEMBL1025020
|
|
Name: Cytotoxicity against mouse P388 cells by MTT assay
Source: ChEMBL
Target: P388
External Id: CHEMBL1025017
|
|
Name: Cytotoxicity against human KB cells by MTT assay
Source: ChEMBL
Target: KB
External Id: CHEMBL1025018
|
|
Name: Agonist activity at LXRalpha (unknown origin) transfected in mouse SN4741 cells at 10...
Source: ChEMBL
Target: Oxysterols receptor LXR-alpha
External Id: CHEMBL2424161
|
|
Name: Binding affinity to Mycobacterium tuberculosis CYP124A1 assessed as type 1 binding mo...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2050041
|
|
Name: Human Liver X receptor-alpha (1H. Liver X receptor-like receptors)
Source: IUPHAR-DB
Target: Liver X receptor-alpha (1H. Liver X receptor-like receptors) [Homo sapiens]
External Id: 602_Human
|
|
Name: Agonist activity at LXRbeta (unknown origin) transfected in mouse SN4741 cells at 10 ...
Source: ChEMBL
Target: Oxysterols receptor LXR-beta
External Id: CHEMBL2424160
|
|
Name: Agonist activity at LXR in mouse SN4741 cells assessed as upregulation of ABCA1 prote...
Source: ChEMBL
Target: N/A
External Id: CHEMBL2424159
|
|
Name: Binding affinity to 2-chloro-5-nitro-N-o-tolylbenzamide-ligated recombinant human N-t...
Source: ChEMBL
Target: Nuclear receptor ROR-gamma
External Id: CHEMBL4714686
|
| cholest-5,24-dien-3β-ol |
| Desmosterol |
| Cholesta-5,24-dien-3b-ol |
| Desmesterol |
| Cholesta-5,24-dien-3-β-ol |
| 3b-Cholesta-5,24-dien-3-ol |
| EINECS 206-236-2 |
| MFCD00056432 |
| 3ß-hydroxy-5,24-cholestadiene |
| Cholesta-5,24-dien-3β-ol (8CI) |
| (3β)-Cholesta-5,24-dien-3-ol |
| (3β,20R)-Cholesta-5,24-dien-3-ol |
| Cholesta-5,24-dien-3β-ol |
| 5,24-cholestadien-3β-ol |
 CAS#:67371-40-8
CAS#:67371-40-8 CAS#:2665-04-5
CAS#:2665-04-5dimethylsilane Structure](https://image.chemsrc.com/caspic/328/220150-72-1.png) CAS#:220150-72-1
CAS#:220150-72-1 CAS#:66414-43-5
CAS#:66414-43-5![2-[(3β)-Cholesta-5,24-dien-3-yloxy]tetrahydro-2H-pyran Structure](https://image.chemsrc.com/caspic/381/51231-31-3.png) CAS#:51231-31-3
CAS#:51231-31-3 CAS#:86476-29-1
CAS#:86476-29-1![(3S,8R,9S,10R,13S,14S,16R,E)-17-ethylidene-10,13-dimethyl-3-((tetrahydro-2H-pyran-2-yl)oxy)-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-16-ol Structure](https://image.chemsrc.com/caspic/434/72777-19-6.png) CAS#:72777-19-6
CAS#:72777-19-6![2-(((3S,8S,9S,10R,13R,14S,17R)-10,13-dimethyl-17-((R)-pent-4-en-2-yl)-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-yl)oxy)tetrahydro-2H-pyran Structure](https://image.chemsrc.com/caspic/235/72727-16-3.png) CAS#:72727-16-3
CAS#:72727-16-3![(3S,8S,9S,10R,13S,14S,16S,17R)-10,13-dimethyl-17-((R)-pent-4-en-2-yl)-3-((tetrahydro-2H-pyran-2-yl)oxy)-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-16-ol Structure](https://image.chemsrc.com/caspic/311/72727-14-1.png) CAS#:72727-14-1
CAS#:72727-14-1![3β-[(tetrahydro-2H-pyran-2-yl)oxy]-16α,17α-epoxy-5-pregnen-20-one Structure](https://image.chemsrc.com/caspic/221/23821-04-7.png) CAS#:23821-04-7
CAS#:23821-04-7 CAS#:474-63-5
CAS#:474-63-5 CAS#:80-97-7
CAS#:80-97-7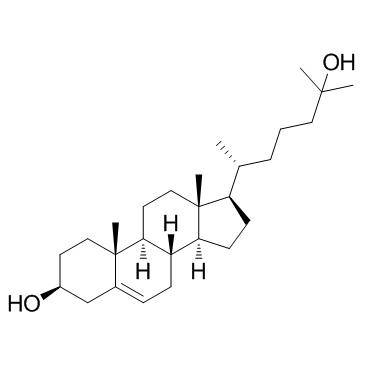 CAS#:2140-46-7
CAS#:2140-46-7 CAS#:77058-74-3
CAS#:77058-74-3