(±)-methyl mandelate
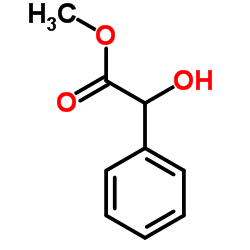
(±)-methyl mandelate structure
|
Common Name | (±)-methyl mandelate | ||
|---|---|---|---|---|
| CAS Number | 4358-87-6 | Molecular Weight | 166.174 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 258.1±0.0 °C at 760 mmHg | |
| Molecular Formula | C9H10O3 | Melting Point | 54-58ºC | |
| MSDS | Chinese USA | Flash Point | 113.1±14.5 °C | |
| Name | Methyl DL-mandelate |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 258.1±0.0 °C at 760 mmHg |
| Melting Point | 54-58ºC |
| Molecular Formula | C9H10O3 |
| Molecular Weight | 166.174 |
| Flash Point | 113.1±14.5 °C |
| Exact Mass | 166.062988 |
| PSA | 46.53000 |
| LogP | 0.90 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.535 |
| Storage condition | -20°C |
| Water Solubility | methanol: 0.1 g/mL, clear |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918199090 |
|---|---|
| Summary | 2918199090 other carboxylic acids with alcohol function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:6.5%。General tariff:30.0% |
|
Hydrolysis of triacetin catalyzed by immobilized lipases: Effect of the immobilization protocol and experimental conditions on diacetin yield
Enzyme Microb. Technol. 48 , 510-517, (2011) The effect of the immobilization protocol and some experimental conditions (pH value and presence of acetonitrile) on the regioselective hydrolysis of triacetin to diacetin catalyzed by lipases has be... |
|
|
Esters of mandelic acid as substrates for (S)-mandelate dehydrogenase from Pseudomonas putida: implications for the reaction mechanism.
Biochemistry 43(7) , 1883-90, (2004) (S)-Mandelate dehydrogenase (MDH) from Pseudomonas putida is a flavin mononucleotide (FMN)-dependent enzyme that oxidizes (S)-mandelate to benzoylformate. In this work, we show that the ethyl and meth... |
|
|
Generation of the enol of methyl mandelate by flash photolysis of methyl phenyldiazoacetate in aqueous solution and study of rates of ketonization of this enol in that medium.
J. Org. Chem. 65(4) , 1175-80, (2000) Flash photolysis of methyl phenyldiazoacetate in aqueous solution produced phenylcarbomethoxycarbene, whose hydration generated a short-lived transient species that was identified as the enol isomer o... |
| EINECS 224-434-7 |
| Hydroxy-phenyl-acetic acid methyl ; ester |
| 3-methoxycarbonyl-2H-cyclohepta<b>furan-2-one |
| Methyl 2-hydroxy-2-phenylacetate |
| Mandelic acid methyl ester |
| 2-methoxycarbonyl-2H-cyclohepta<b>furan-2-one |
| methyl-2-hydroxy-2-phenylacetate |
| (±)-methyl mandelate |
| methyl mandelate |
| methyl-2H-cyclohepta<b>furan-2-one-3-carboxylate |
| QYR&VO1 |
| MFCD00004493 |
| hydroxyphenylacetic acid methyl ester |
| Methyl DL-Mandelate |
| Methyl hydroxy(phenyl)acetate |
| DL-Mandelic Acid Methyl Ester |
| Benzeneacetic acid, α-hydroxy-, methyl ester |
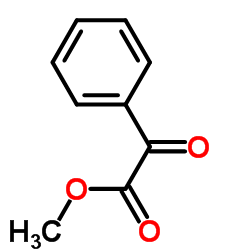 CAS#:15206-55-0
CAS#:15206-55-0 CAS#:124-38-9
CAS#:124-38-9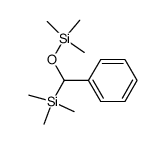 CAS#:53172-92-2
CAS#:53172-92-2 CAS#:18107-18-1
CAS#:18107-18-1 CAS#:67-56-1
CAS#:67-56-1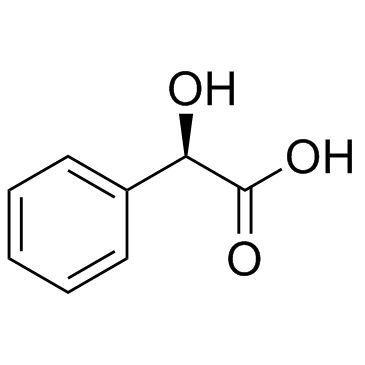 CAS#:611-71-2
CAS#:611-71-2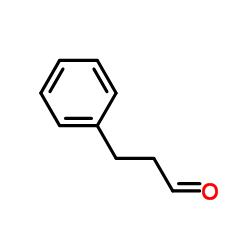 CAS#:104-53-0
CAS#:104-53-0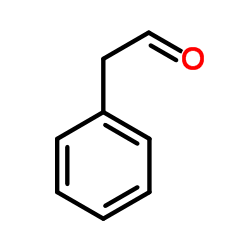 CAS#:122-78-1
CAS#:122-78-1 CAS#:1074-12-0
CAS#:1074-12-0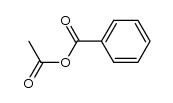 CAS#:2819-08-1
CAS#:2819-08-1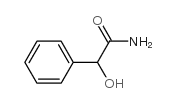 CAS#:4410-31-5
CAS#:4410-31-5 CAS#:451-40-1
CAS#:451-40-1 CAS#:515-30-0
CAS#:515-30-0 CAS#:939-55-9
CAS#:939-55-9 CAS#:4335-77-7
CAS#:4335-77-7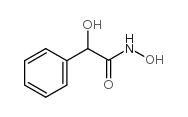 CAS#:2292-53-7
CAS#:2292-53-7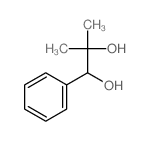 CAS#:20907-13-5
CAS#:20907-13-5 CAS#:19020-59-8
CAS#:19020-59-8
