clemizole
Modify Date: 2025-08-21 01:18:38
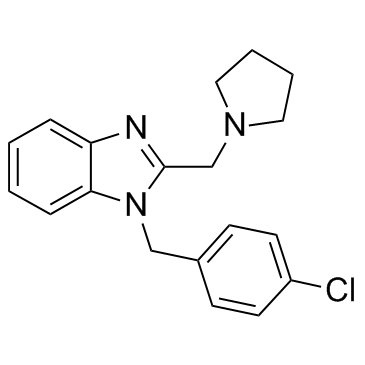
clemizole structure
|
Common Name | clemizole | ||
|---|---|---|---|---|
| CAS Number | 442-52-4 | Molecular Weight | 325.835 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 506.1±40.0 °C at 760 mmHg | |
| Molecular Formula | C19H20ClN3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 259.9±27.3 °C | |
Use of clemizoleClemizole is an H1 histamine receptor antagonist, is found to substantially inhibit HCV replication. The IC50 of Clemizole for RNA binding by NS4B is 24±1 nM, whereas its EC50 for viral replication is 8 µM. |
| Name | clemizole |
|---|---|
| Synonym | More Synonyms |
| Description | Clemizole is an H1 histamine receptor antagonist, is found to substantially inhibit HCV replication. The IC50 of Clemizole for RNA binding by NS4B is 24±1 nM, whereas its EC50 for viral replication is 8 µM. |
|---|---|
| Related Catalog | |
| Target |
IC50: 24 nM (NS4B)[1] H1 histamine receptor[1] |
| In Vitro | Clemizole hydrochloride is found to inhibit HCV RNA replication in cell culture that is mediated by its suppression of NS4B’s RNA binding, with little toxicity for the host cell. The EC50 of Clemizole on the W55R mutant J6/JFH RNA is ~18 µM (2.25 times the EC50 of the wild-type RNA)[1]. Clemizole is a novel inhibitor of TRPC5 channels. Clemizole efficiently blocks TRPC5 currents and Ca2+ entry in the low micromolar range (IC50=1.0-1.3 µM). Clemizole exhibits a six-fold selectivity for TRPC5 over TRPC4β (IC50=6.4 µM), the closest structural relative of TRPC5, and an almost 10-fold selectivity over TRPC3 (IC50=9.1 µM) and TRPC6 (IC50=11.3 µM). Clemizole hydrochloride as a novel blocker of TRPC5 with a half-maximal inhibitory concentration of 1.1 µM. The concentration-response curves confirmed a concentration-dependent block of TRPC5 by Clemizole and revealed an apparent IC50 of 1.1±0.04 µM[2]. |
| In Vivo | Clemizole hydrochloride has an unexpectedly short plasma half-life (measured at 0.15 hours); it is very rapidly biotransformed into a glucuronide (M14) and a dealkylated metabolite (M12) and into a variety of lesser metabolites in C57BL/6J mice[3]. |
| Cell Assay | Huh7.5 cells are maintained in DMEM supplemented with 1% L-glutamine, 1% penicillin, 1% streptomycin, 1× nonessential amino acids and 10% FBS. Cell lines are passaged twice weekly after treatment with 0.05% trypsin-0.02% EDTA and seeding at a dilution of 1:5. Subconfluent Huh7.5 cells are trypsinized and collected by centrifugation at 700g for 5 min. The cells are then washed three times in ice-cold RNase-free PBS and resuspended at 1.5×107 cells/mL in PBS. Wild-type or mutant FL-J6/JFH-5′C19Rluc2AUbi RNA for electroporation is generated by transcription of XbaI linearized DNA templates using the T7 MEGAscript kit, followed by purification (RNA transcription and fluorescent labeling). We mixed 5 µg of RNA with 400 µL of washed Huh7.5 cells in a 2-mm-gap cuvette (BTX) and immediately pulsed (0.82 kV, five 99 µs pulses) with a BTX-830 electroporator. After a 10 min recovery at 25°C, pulsed cells are diluted into 10 mL of prewarmed growth medium. Cells from several electroporations are pooled to a common stock and seeded in 6-well plates (5×105 cells per well). After 24 h, medium is replaced and cells are grown in the presence of serial dilutions of the various inhibitory compounds (e.g., Clemizole hydrochloride) identified in the screen. Seventeen commercially available compounds, out of the 18 identified, are analyzed. Untreated cells are used as a negative control for water-soluble compounds. For compounds (e.g., Clemizole hydrochloride) solubilized in DMSO, untreated cells are grown in the presence of corresponding concentrations of the solvent as a negative control. Medium is changed daily. After 72 h of treatment cells are subjected to an Alamar Blue-based viability assay and luciferase assay. After 72 h of treatment cells are incubated for 3 h at 37°C in the presence of 10% Alamar Blue reagent. Plates are then scanned and fluorescence is detected by using FLEXstation II 384. Depending on the inhibitory compound’s solvent (e.g., Clemizole hydrochloride), water or DMSO, signal is normalized relatively to untreated samples or samples grown in the presence of DMSO, respectively[1]. |
| Animal Admin | Mice[3] Eight control NOG mice and eight humanized TK-NOG mice are administered 25 mg/kg by mouth Clemizole, and blood samples are collected 30 minutes after administration. The C57BL/6J mice (3 per time point) are given 25 mg/kg by mouth clemizole, and blood samples are collected for analysis at 15 and 30 minutes and 1, 2, 4, and 6 hours after administration. For the DDI studies, eight humanized TK-NOG mice are given Clemizole (25 mg/kg by mouth) with or without Ritonavir (20 mg/kg by mouth), and blood samples are collected 30 minutes after administration. Six of these mice are also treated with Debrisoquine (10 mg/kg by mouth) in the presence or absence of Ritonavir (20 mg/kg by mouth), and plasma samples are obtained 2 hours later for analysis. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 506.1±40.0 °C at 760 mmHg |
| Molecular Formula | C19H20ClN3 |
| Molecular Weight | 325.835 |
| Flash Point | 259.9±27.3 °C |
| Exact Mass | 325.134583 |
| PSA | 21.06000 |
| LogP | 5.10 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.656 |
| InChIKey | CJXAEXPPLWQRFR-UHFFFAOYSA-N |
| SMILES | Clc1ccc(Cn2c(CN3CCCC3)nc3ccccc32)cc1 |
| Storage condition | -20°C |
| HS Code | 2933990090 |
|---|
| Precursor 10 | |
|---|---|
| DownStream 0 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
Total 256, Current Page 1 of 26
1
2
3
4
5
| Histakool |
| 1-(4-Chlorobenzyl)-2-(1-pyrrolidinylmethyl)-1H-benzimidazole |
| Clemizol |
| 1-(p-Chlorobenzyl)-2-(1-pyrrolidinylmethyl)benzimidazole |
| 1-[(4-Chlorophenyl)methyl]-2-(pyrrolidinylmethyl)-1H-benzimidazole |
| clemizole |
| 1-(4-chloro-benzyl)-2-pyrrolidin-1-ylmethyl-1H-benzoimidazole |
| 1-(4-Chlorobenzyl)-2-(pyrrolidin-1-ylmethyl)-1H-benzimidazole |
| Histacuran |
| Klemidox |
| 1-(4-Chlorbenzyl)-2-(pyrrolidin-1-ylmethyl)-1H-benzimidazol |
| Allercur |
| 1-(p-Chlorobenzyl)-2-pyrrolidylmethylenebenzimidazole |
| 1-(4-chloro-benzyl)-2-pyrrolidinomethyl-1H-benzimidazole |
| Reactrol |
| 1-(4-Chlor-benzyl)-2-pyrrolidinomethyl-1H-benzimidazol |
| 1H-Benzimidazole,1-[(4-chlorophenyl)methyl]-2-(1-pyrrolidinylmethyl) |
| histacur |
| 1-[(4-chlorophenyl)methyl]-2-(pyrrolidin-1-ylmethyl)benzimidazole |
| Clemizolum |
| clemizolum [INN_la] |
 CAS#:123-75-1
CAS#:123-75-1 CAS#:7189-18-6
CAS#:7189-18-6 CAS#:622-95-7
CAS#:622-95-7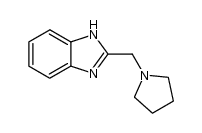 CAS#:2948-92-7
CAS#:2948-92-7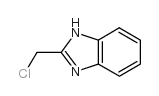 CAS#:4857-04-9
CAS#:4857-04-9![pyrrolidino-acetic acid-[2-(4-chloro-benzylamino)-anilide] Structure](https://image.chemsrc.com/caspic/169/718634-79-8.png) CAS#:718634-79-8
CAS#:718634-79-8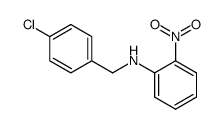 CAS#:5822-16-2
CAS#:5822-16-2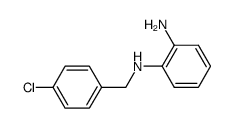 CAS#:5729-18-0
CAS#:5729-18-0![pyrrolidino-acetic acid-[2-(4-chloro-benzylidenamino)-anilide] Structure](https://image.chemsrc.com/caspic/367/109037-60-7.png) CAS#:109037-60-7
CAS#:109037-60-7 CAS#:104-86-9
CAS#:104-86-9