3-Aminopyridine
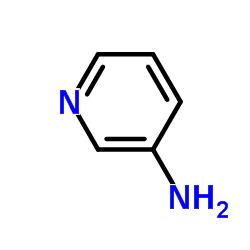
3-Aminopyridine structure
|
Common Name | 3-Aminopyridine | ||
|---|---|---|---|---|
| CAS Number | 462-08-8 | Molecular Weight | 94.115 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 251.0±0.0 °C at 760 mmHg | |
| Molecular Formula | C5H6N2 | Melting Point | 60-63 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 126.0±7.0 °C | |
| Symbol |


GHS06, GHS08 |
Signal Word | Danger | |
| Name | 3-Aminopyridine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 251.0±0.0 °C at 760 mmHg |
| Melting Point | 60-63 °C(lit.) |
| Molecular Formula | C5H6N2 |
| Molecular Weight | 94.115 |
| Flash Point | 126.0±7.0 °C |
| Exact Mass | 94.053101 |
| PSA | 38.91000 |
| LogP | -0.02 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.588 |
| InChIKey | CUYKNJBYIJFRCU-UHFFFAOYSA-N |
| SMILES | Nc1cccnc1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |


GHS06, GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H311-H315-H319-H331-H335-H373 |
| Precautionary Statements | P261-P280-P301 + P310-P305 + P351 + P338-P311 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T:Toxic |
| Risk Phrases | R25;R36/37/38 |
| Safety Phrases | S36/37/39-S45-S37/39-S28B-S26 |
| RIDADR | UN 2671 6.1/PG 2 |
| WGK Germany | 3 |
| RTECS | US1650000 |
| Packaging Group | II |
| Hazard Class | 6.1 |
| HS Code | 2933399090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Anharmonic vibrational analysis of 3,4-diaminopyridine and 3-aminopyridine by density functional theory calculations.
Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 76(5) , 502-12, (2010) In this work, we will report a combined experimental and theoretical study on molecular structure and vibrational analysis of 3,4-diaminopyridine (3,4-DAP) and 3-aminopyridine (3-AP). The Fourier tran... |
|
|
Evaluation of electrophilic heteroaromatic substitution: synthesis of heteroaromatic-fused pyrimidine derivatives via sequential three-component heterocyclization.
J. Org. Chem. 77(19) , 8492-500, (2012) A new sequential three-component heterocyclization was developed by reacting aromatic and heterocyclic substrates, including aminobenzenes, 1-aminonaphthalene, 2-aminopyrazines, 5-aminopyrazoles, 3-am... |
|
|
The significance of the temporal bond polarizabilty relaxation of 2- and 3-aminopyridine by 514.5 nm excitation for the nonresonant Raman virtual states.
Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 71(4) , 1588-93, (2008) Our algorithm [B. Tian, G. Wu, G. Liu, J. Chem. Phys. 87 (1987) 7300] is introduced to obtain the temporal bond polarizabilities of 2- and 3-aminopyridine from their Raman intensities, which supply fr... |
| B-PYRIDYLAMINE |
| 5-amino pyridine |
| pyridin-3-ylamine |
| 3-amino-pyridin |
| T6NJ CZ |
| pyridine-3-amine |
| 3APY |
| 3-amino-pyridine |
| 3-Aminopyridine |
| m-Aminopyridine |
| 3-pyridinylamine |
| 3-Pyridinamine |
| pyridin-3-amine |
| EINECS 207-322-2 |
| 3-PYRIDYLAMINE |
| 3-aminepyridin |
| MFCD00006400 |
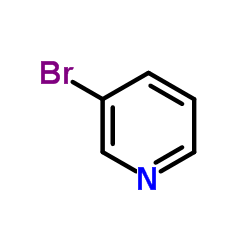 CAS#:626-55-1
CAS#:626-55-1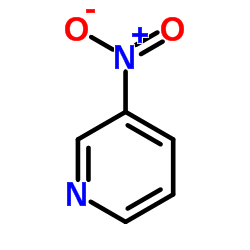 CAS#:2530-26-9
CAS#:2530-26-9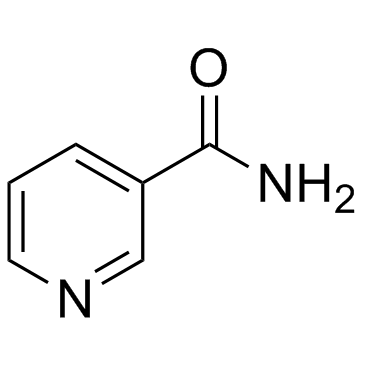 CAS#:98-92-0
CAS#:98-92-0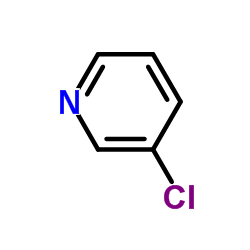 CAS#:626-60-8
CAS#:626-60-8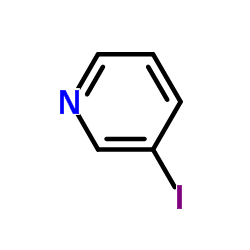 CAS#:1120-90-7
CAS#:1120-90-7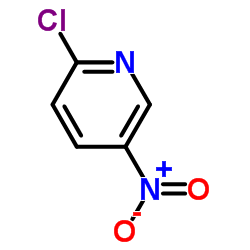 CAS#:4548-45-2
CAS#:4548-45-2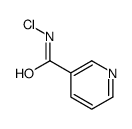 CAS#:63458-55-9
CAS#:63458-55-9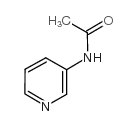 CAS#:5867-45-8
CAS#:5867-45-8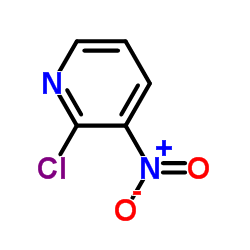 CAS#:5470-18-8
CAS#:5470-18-8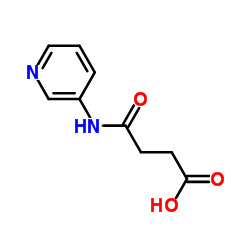 CAS#:25604-13-1
CAS#:25604-13-1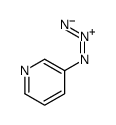 CAS#:10296-29-4
CAS#:10296-29-4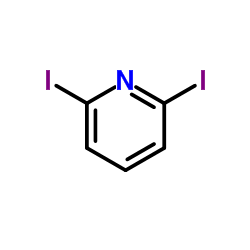 CAS#:53710-17-1
CAS#:53710-17-1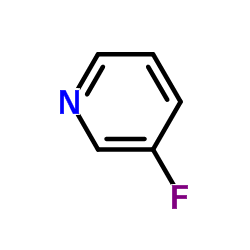 CAS#:372-47-4
CAS#:372-47-4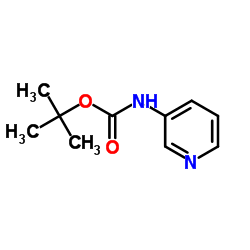 CAS#:56700-70-0
CAS#:56700-70-0 CAS#:3256-88-0
CAS#:3256-88-0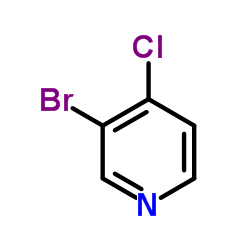 CAS#:36953-42-1
CAS#:36953-42-1 CAS#:5423-54-1
CAS#:5423-54-1 CAS#:329214-79-1
CAS#:329214-79-1 CAS#:54247-51-7
CAS#:54247-51-7
