5-Hydroxyfisetin
Modify Date: 2025-08-23 17:29:04
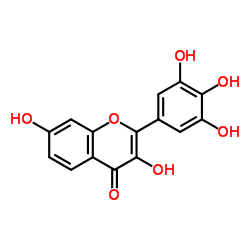
5-Hydroxyfisetin structure
|
Common Name | 5-Hydroxyfisetin | ||
|---|---|---|---|---|
| CAS Number | 490-31-3 | Molecular Weight | 302.236 | |
| Density | 1.8±0.1 g/cm3 | Boiling Point | 669.9±55.0 °C at 760 mmHg | |
| Molecular Formula | C15H10O7 | Melting Point | 326-328ºC | |
| MSDS | N/A | Flash Point | 258.6±25.0 °C | |
Use of 5-HydroxyfisetinRobinetin (3,3',4',5',7-Pentahydroxyflavone), a naturally occurring flavonoid with remarkable ‘two color’ intrinsic fluorescence properties, has antifungal, antiviral, antibacterial, antimutagenesis, and antioxidant activity. Robinetin also can inhibit lipid peroxidation and protein glycosylation[1][2][3][4][5]. |
| Name | robinetin |
|---|---|
| Synonym | More Synonyms |
| Description | Robinetin (3,3',4',5',7-Pentahydroxyflavone), a naturally occurring flavonoid with remarkable ‘two color’ intrinsic fluorescence properties, has antifungal, antiviral, antibacterial, antimutagenesis, and antioxidant activity. Robinetin also can inhibit lipid peroxidation and protein glycosylation[1][2][3][4][5]. |
|---|---|
| Related Catalog | |
| In Vitro | Robinetin (0.1-10 μM; 1 h) inhibits HIV integrase cleavage and integration in a dose-dependent manner[1]. Robinetin inhibits the DNA synthesis in Proteus vulgaris, and the RNA synthesis in S. aureus[2]. Robinetin (100-200 or 25 μM; 1 or 72 h) inhibits egg yolk phosphatidylcholine (EYPC) membrane lipid peroxidation and hemoglobin A (HbA) glycosylation with high efficiency[3]. Robinetin exhibits photo-induced excited-state intramolecular proton transfer resulting in ‘two color’ (in ‘blue-violet’ and ‘yellow-green’ regions) fluorescence characteristic of flavonols, the relative contributions between the two colors being strongly modulated by the local environment of the fluorophore[3]. |
| References |
| Density | 1.8±0.1 g/cm3 |
|---|---|
| Boiling Point | 669.9±55.0 °C at 760 mmHg |
| Melting Point | 326-328ºC |
| Molecular Formula | C15H10O7 |
| Molecular Weight | 302.236 |
| Flash Point | 258.6±25.0 °C |
| Exact Mass | 302.042664 |
| PSA | 131.36000 |
| LogP | 2.55 |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.823 |
| InChIKey | SOEDEYVDCDYMMH-UHFFFAOYSA-N |
| SMILES | O=c1c(O)c(-c2cc(O)c(O)c(O)c2)oc2cc(O)ccc12 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
| Precursor 9 | |
|---|---|
| DownStream 0 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
Total 42, Current Page 1 of 5
1
2
3
4
5
| Robinetin |
| Norkanugin |
| 3,7,3',4',5'-pentahydroxyflavonol |
| 3,7,3',4',5'-pentahydroxyflavone |
| 3,7-Dihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-chromen-4-one |
| 3,7-dihydroxy-2-(3,4,5-trihydroxyphenyl)chromen-4-one |
| 5-Hydroxyfisetin |
| EINECS 207-709-6 |
| 3,3',4',5',7-Pentahydroxyflavone |
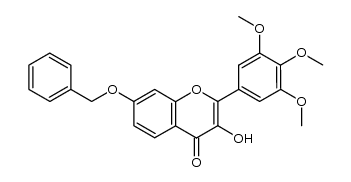 CAS#:354119-95-2
CAS#:354119-95-2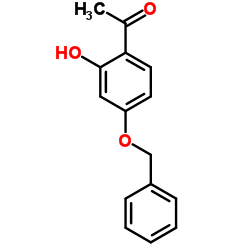 CAS#:29682-12-0
CAS#:29682-12-0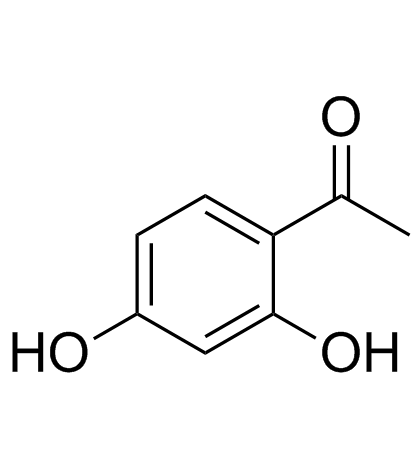 CAS#:89-84-9
CAS#:89-84-9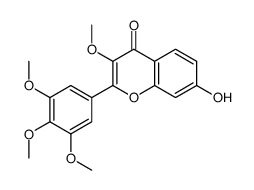 CAS#:20979-43-5
CAS#:20979-43-5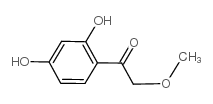 CAS#:57280-75-8
CAS#:57280-75-8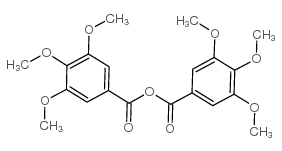 CAS#:1719-88-6
CAS#:1719-88-6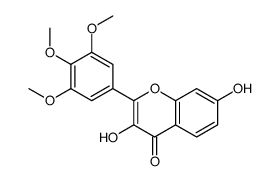 CAS#:132594-09-3
CAS#:132594-09-3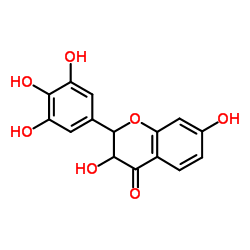 CAS#:4382-33-6
CAS#:4382-33-6 CAS#:109980-57-6
CAS#:109980-57-6
