Isonipecotic acid

Isonipecotic acid structure
|
Common Name | Isonipecotic acid | ||
|---|---|---|---|---|
| CAS Number | 498-94-2 | Molecular Weight | 129.157 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 265.8±33.0 °C at 760 mmHg | |
| Molecular Formula | C6H11NO2 | Melting Point | >300 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 114.5±25.4 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Isonipecotic acidIsonipecotic acid is a GABAA receptor partial agonist[1]. |
| Name | Isonipecotic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Isonipecotic acid is a GABAA receptor partial agonist[1]. |
|---|---|
| Related Catalog | |
| Target |
GABAA Receptor[1] |
| In Vitro | Isonipecotic acid inhibits [3H]GABA binding (2°C) with an IC50 of 0.33 μM[2]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 265.8±33.0 °C at 760 mmHg |
| Melting Point | >300 °C(lit.) |
| Molecular Formula | C6H11NO2 |
| Molecular Weight | 129.157 |
| Flash Point | 114.5±25.4 °C |
| Exact Mass | 129.078979 |
| PSA | 49.33000 |
| LogP | -0.08 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.479 |
| InChIKey | SRJOCJYGOFTFLH-UHFFFAOYSA-N |
| SMILES | O=C(O)C1CCNCC1 |
| Storage condition | Store at 0-5°C |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NS5150000 |
| HS Code | 2942000000 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain canc... |
|
|
Genetic mapping of targets mediating differential chemical phenotypes in Plasmodium falciparum.
Nat. Chem. Biol. 5 , 765-71, (2009) Studies of gene function and molecular mechanisms in Plasmodium falciparum are hampered by difficulties in characterizing and measuring phenotypic differences between individual parasites. We screened... |
|
|
Three-dimensional quantitative structure-activity relationship analyses of substrates of the human proton-coupled amino acid transporter 1 (hPAT1).
Bioorg. Med. Chem. 19 , 6409-18, (2011) The proton-coupled amino acid transporter hPAT1 has recently gained much interest due to its ability to transport small drugs thereby allowing their oral administration. A three-dimensional quantitati... |
| Isonipecotic acid |
| 4-Hexahydroisonicotinic acid |
| Piperidine-4-carboxylic acid |
| UNII-M5TZP1RWIE |
| 4-Piperidinecarboxylic acid |
| Hexahydroisonicotinic acid |
| EINECS 207-872-3 |
| MFCD00006004 |
| Isonipecoticacid |
 CAS#:13602-12-5
CAS#:13602-12-5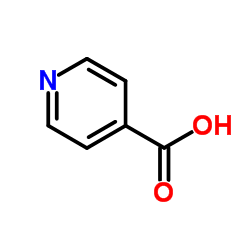 CAS#:55-22-1
CAS#:55-22-1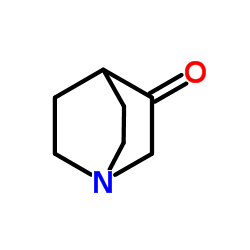 CAS#:3731-38-2
CAS#:3731-38-2 CAS#:10314-98-4
CAS#:10314-98-4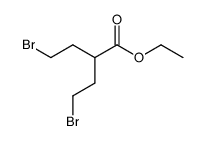 CAS#:857778-39-3
CAS#:857778-39-3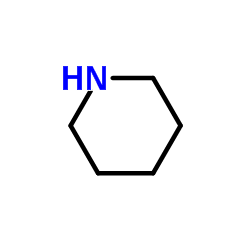 CAS#:110-89-4
CAS#:110-89-4 CAS#:64-18-6
CAS#:64-18-6 CAS#:6238-69-3
CAS#:6238-69-3![1-[bis(dimethylamino)methylene]piperidinium 4-carboxylate Structure](https://image.chemsrc.com/caspic/331/1219594-56-5.png) CAS#:1219594-56-5
CAS#:1219594-56-5 CAS#:5398-44-7
CAS#:5398-44-7 CAS#:101264-48-6
CAS#:101264-48-6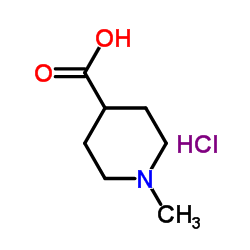 CAS#:71985-80-3
CAS#:71985-80-3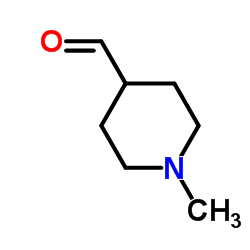 CAS#:50675-21-3
CAS#:50675-21-3 CAS#:5274-99-7
CAS#:5274-99-7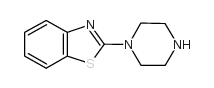 CAS#:55745-83-0
CAS#:55745-83-0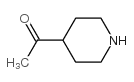 CAS#:30818-11-2
CAS#:30818-11-2 CAS#:55695-51-7
CAS#:55695-51-7 CAS#:92822-02-1
CAS#:92822-02-1![2-(Piperidin-4-yl)benzo[d]thiazole structure](https://image.chemsrc.com/caspic/116/51784-73-7.png) CAS#:51784-73-7
CAS#:51784-73-7 CAS#:51784-03-3
CAS#:51784-03-3
