Pronethalol hydrochloride
Modify Date: 2025-08-25 10:58:03
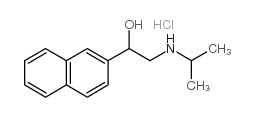
Pronethalol hydrochloride structure
|
Common Name | Pronethalol hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 51-02-5 | Molecular Weight | 265.77800 | |
| Density | 1.074g/cm3 | Boiling Point | 322.4ºC at 760mmHg | |
| Molecular Formula | C15H20ClNO | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 84.3ºC | |
Use of Pronethalol hydrochloridePronethalol ((±)-Pronethalo) is a non-selective β-adrenergic antagonist. Pronethalol is a potent inhibitor of Sox2 expression. Pronethalol protects against and to reverse Digitalis-induced ventricular arrhythmias, and limits the cerebral arteriovenous malformation (AVMs)[1][2]. |
| Name | 1-naphthalen-2-yl-2-(propan-2-ylamino)ethanol,hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Pronethalol ((±)-Pronethalo) is a non-selective β-adrenergic antagonist. Pronethalol is a potent inhibitor of Sox2 expression. Pronethalol protects against and to reverse Digitalis-induced ventricular arrhythmias, and limits the cerebral arteriovenous malformation (AVMs)[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Pronethalol (2, 10, 20 μM) represses EGFP expression in a dose- and time-dependent manner in ReNcell VM cells. Pronethalol (10 μM; 2 days) reduces Sox2 expression to less than 10% after 2 days of treatment[2]. |
| In Vivo | Pronethalol (0.15 mg/g; daily; for 14 days) stabilizes endothelial differentiation, lumen formation and improves cerebral arteriovenous malformation (AVMs) in Mgp–/– mice[2]. |
| References |
| Density | 1.074g/cm3 |
|---|---|
| Boiling Point | 322.4ºC at 760mmHg |
| Molecular Formula | C15H20ClNO |
| Molecular Weight | 265.77800 |
| Flash Point | 84.3ºC |
| Exact Mass | 265.12300 |
| PSA | 32.26000 |
| LogP | 4.06410 |
| Index of Refraction | 1.596 |
| InChIKey | QONLGXRPRAIDGI-UHFFFAOYSA-N |
| SMILES | CC(C)NCC(O)c1ccc2ccccc2c1.Cl |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| HS Code | 2922199090 |
|---|
| HS Code | 2922199090 |
|---|---|
| Summary | 2922199090. other amino-alcohols, other than those containing more than one kind of oxygen function, their ethers and esters; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Tocris HTS for Inhibitors of Aerobactin Synthetase lucA
Source: 23265
External Id: IucA Pilot Assay Tocris Library
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
Total 130, Current Page 1 of 13
1
2
3
4
5
| PRONETHALOL HYDROCHLORIDE |
| Pronetalol hydrochloride |
| Nethalide hydrochloride |
| DL-Pronethalol hydrochloride |
| Pronethalol.HCl |
| Naphthylisoproterenol hydrochloride |
| Alderlin hydrochloride |