Flunarizine
Modify Date: 2025-08-25 11:49:45
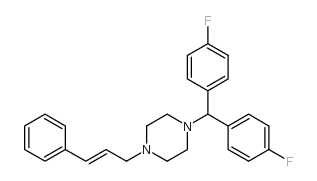
Flunarizine structure
|
Common Name | Flunarizine | ||
|---|---|---|---|---|
| CAS Number | 52468-60-7 | Molecular Weight | 404.49500 | |
| Density | 1.17 g/cm3 | Boiling Point | 511.3ºC at 760 mmHg | |
| Molecular Formula | C26H26F2N2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of FlunarizineFlunarizine is a potent dual Na+/Ca2+ channel (T-type) blocker. Flunarizine is a D2 dopamine receptor antagonist. Flunarizine shows anticonvulsive and antimigraine activity, and peripheral vasodilator effects[1][2][3][4][5]. |
| Name | Flunarizine |
|---|---|
| Synonym | More Synonyms |
| Description | Flunarizine is a potent dual Na+/Ca2+ channel (T-type) blocker. Flunarizine is a D2 dopamine receptor antagonist. Flunarizine shows anticonvulsive and antimigraine activity, and peripheral vasodilator effects[1][2][3][4][5]. |
|---|---|
| Related Catalog | |
| Target |
D2 Receptor |
| In Vitro | Flunarizine blocks sodium currents (INa) and calcium currents (ICa) with IC50 values of 0.94 μM and 1.77 μM in cultured rat cortical neurons, respectively[2]. Flunarizine (10 and 30 μM; 24 h) shows cytotoxic effects to chromaffin cells[4]. Flunarizine (1-30 μM) causes clear cytoprotection of chromaffin cell at concentrations of 3-10 μM[4]. Cell Cytotoxicity Assay[4] Cell Line: Chromaffin cells[4] Concentration: 10 and 30 μM Incubation Time: 24 hours Result: Showed a tendency to increase cell death at the concentration of 10 μM, and showed near 100% cell loss at the concentration of 30 μM. |
| In Vivo | Flunarizine (intraperitoneal injection; 30 mg/kg; once) protects mice from lipopolysaccharide- (LPS-) induced acute lung injury (ALI)[5]. Animal Model: Male BALB/c mice (6-8 weeks old) with acute lung injury induced by lipopolysaccharide[5] Dosage: 30 mg/kg Administration: Intraperitoneal injection; 30 mg/kg; once Result: Suppressed the LPS-induced cell influx, protein leakage, and inflammatory cytokines release. Inhibited the pulmonary inflammation. |
| Density | 1.17 g/cm3 |
|---|---|
| Boiling Point | 511.3ºC at 760 mmHg |
| Molecular Formula | C26H26F2N2 |
| Molecular Weight | 404.49500 |
| Exact Mass | 404.20600 |
| PSA | 6.48000 |
| LogP | 5.26100 |
| Index of Refraction | 1.606 |
| InChIKey | SMANXXCATUTDDT-QPJJXVBHSA-N |
| SMILES | Fc1ccc(C(c2ccc(F)cc2)N2CCN(CC=Cc3ccccc3)CC2)cc1 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Precursor 2 | |
|---|---|
| DownStream 0 | |
Total 564, Current Page 1 of 57
1
2
3
4
5
| flunarazine |
| (E)-1-[bis(4-fluorophenyl)methyl]-4-(cinnamyl)piperazine |
| Sibelium |
| 1-[bis(4-fluorophenyl)methyl]-4-cinnamylpiperazine |
| 1-[Bis(4-fluorophenyl)methyl]-4-[(2E)-3-phenyl-2-propenyl]piperazine |
| EINECS 257-937-5 |
| (E)-1-[Bis-(p-fluorophenyl)methyl]-4-cinnamylpiperazine |
| Fluarizine |
| (e)-piperazin |
| 1-[Bis(4-fluorophenyl)methyl]-4-[(E)-3-phenyl-2-propenyl]piperazine |
| 1-[bis(4-fluorophenyl)methyl]-4-(3-phenyl-2-propenyl)piperazine |
| (e)-1-[bis(4-fluorophenyl)methyl]-4-(3-phenyl-2-propenyl)piperazine |
| MFCD00242731 |
| Piperazine,1-[bis(4-fluorophenyl)methyl]-4-(3-phenyl-2-propenyl)-,(E) |