| Description |
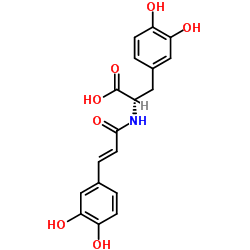
Clovamide (trans-Clovamide), a natural phenolic compound, is a potent antioxidant. Clovamide is an excellent ROS and oxygen radical scavenger. Clovamide also has anti-inflammatory and neuroprotective effects[1][2]. Clovamide is an anti-microbial with activity against the human pathogens influenza A subtype H5N1, Trypanosoma evansi, and Heliobacter pylori[3].
|
| Related Catalog |
|
| In Vitro |
Clovamide is able to protect neurons from injury in three in vitro models of neuronal death: oxidative stress, excitotoxicity and OGD/reoxygenation. In SH-SY5Y human neuroblastoma cells, Clovamide (10-100 µM) significantly protects cell death, with an EC50 value of 3.6 µM. Clovamide also significantly enhances PPARγ expression[2]. Clovamide inhibits growth of three pathogens of cacao in the genus Phytophthora, is a substrate for cacao polyphenol oxidase, and is a contributor to enzymatic browning. Clovamide inhibiteds proteinase and pectinase in vitro[3].
|
| References |
[1]. Naike Ye, et al. Antioxidant studies by hydrodynamic voltammetry and DFT, quantitative analyses by HPLC-DAD of clovamide, a natural phenolic compound found in Theobroma Cacao L. beans. Food Chem. 2021 Mar 30;341(Pt 2):128260. [2]. S Fallarini, et al. Clovamide and rosmarinic acid induce neuroprotective effects in in vitro models of neuronal death. Br J Pharmacol. 2009 Jul;157(6):1072-84.
|