Hordenine
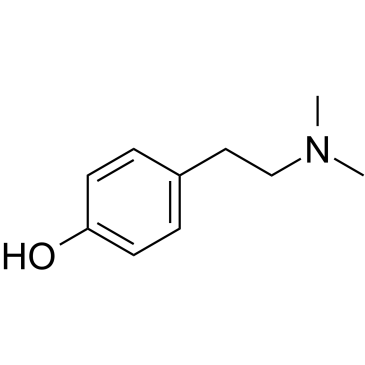
Hordenine structure
|
Common Name | Hordenine | ||
|---|---|---|---|---|
| CAS Number | 539-15-1 | Molecular Weight | 165.232 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 270.2±23.0 °C at 760 mmHg | |
| Molecular Formula | C10H15NO | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 123.5±21.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of HordenineHordenine, an alkaloid found in plants, inhibits melanogenesis by suppression of cyclic adenosine monophosphate (cAMP) production[1]. |
| Name | hordenine |
|---|---|
| Synonym | More Synonyms |
| Description | Hordenine, an alkaloid found in plants, inhibits melanogenesis by suppression of cyclic adenosine monophosphate (cAMP) production[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 270.2±23.0 °C at 760 mmHg |
| Molecular Formula | C10H15NO |
| Molecular Weight | 165.232 |
| Flash Point | 123.5±21.3 °C |
| Exact Mass | 165.115356 |
| PSA | 23.47000 |
| LogP | 1.40 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.542 |
| InChIKey | KUBCEEMXQZUPDQ-UHFFFAOYSA-N |
| SMILES | CN(C)CCc1ccc(O)cc1 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H317-H319 |
| Precautionary Statements | P280-P305 + P351 + P338 |
| Hazard Codes | Xn |
| Risk Phrases | 22-36-43 |
| Safety Phrases | 26-36/37 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2922199090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922199090 |
|---|---|
| Summary | 2922199090. other amino-alcohols, other than those containing more than one kind of oxygen function, their ethers and esters; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Development of immunoassays for tyramine and tryptamine toxins of Phalaris aquatica L.
J. Agric. Food Chem. 48(1) , 27-32, (2000) The leaves of the perennial pasture grass Phalaris aquatica L. (phalaris) contain two groups of known toxins, indole alkaloids, primarily dimethyltryptamines and N-methyltyramines, which cause illness... |
|
|
Some hematological and histopathological effects of the alkaloids gramine and hordenine on meadow voles (Microtus pennsylvanicus).
Toxicology 18(2) , 125-31, (1980) Meadow voles (Microtus pennsylvanicus) were used to evaluate the relative toxicity of the alkaloids, gramine and hordenine, which are present in reed canarygrass (Phalaris arundinacea) and to assess t... |
|
|
Rapid formation of N-nitrosodimethylamine from gramine, a naturally occurring precursor in barley malt.
IARC Sci. Publ. (57) , 337-46, (1984) The two tertiary amine alkaloids, hordenine and gramine, which are biosynthesized in malt during germination, were subjected to nitrosation under conditions typical for the study of tertiary amine nit... |
| 4-[2-(Dimethylamino)ethyl]phenol |
| Cactine |
| Anhaline |
| Peyocactine |
| N,N-dimethyltyramine |
| Hordenin |
| Hordetin |
| Hordenine |
| p-[2-(Dimethylamino)ethyl]phenol |
| 2-(4-Hydroxyphenyl)-N,N-dimethylethylamine (4-(2-Dimethylaminoethyl)phenol |
| MFCD00051462 |
| p-Hydroxy-N,N-dimethylphenethylamine |
| 4-(2-(Dimethylamino)ethyl)phenol |
| Ordenina |
| Eremursine |
| EINECS 222-740-5 |
| Anhalin |
| Ordenine |
| 4-Hydroxy-N,N-dimethylphenethylamine |
| N,N-Dimethyl-p-hydroxyphenethylamine |
| Anhalin-d6 |
| N,N-dimethyl-2-(4-hydroxyphenyl)ethylamine |
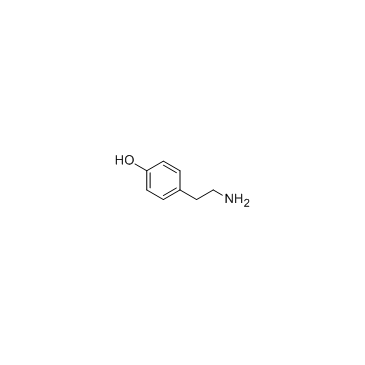 CAS#:51-67-2
CAS#:51-67-2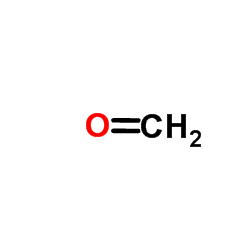 CAS#:50-00-0
CAS#:50-00-0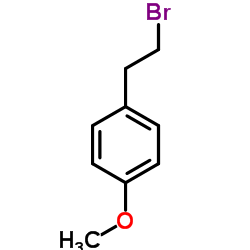 CAS#:14425-64-0
CAS#:14425-64-0 CAS#:124-40-3
CAS#:124-40-3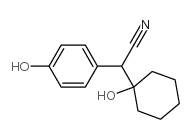 CAS#:918344-20-4
CAS#:918344-20-4 CAS#:14140-15-9
CAS#:14140-15-9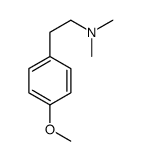 CAS#:775-33-7
CAS#:775-33-7 CAS#:104-47-2
CAS#:104-47-2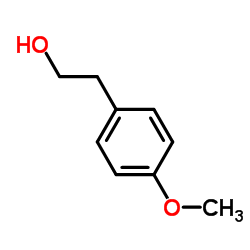 CAS#:702-23-8
CAS#:702-23-8 CAS#:55-81-2
CAS#:55-81-2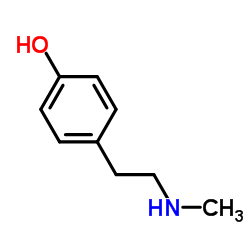 CAS#:370-98-9
CAS#:370-98-9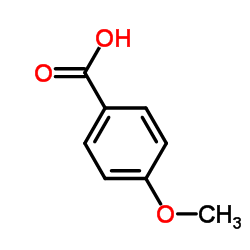 CAS#:100-09-4
CAS#:100-09-4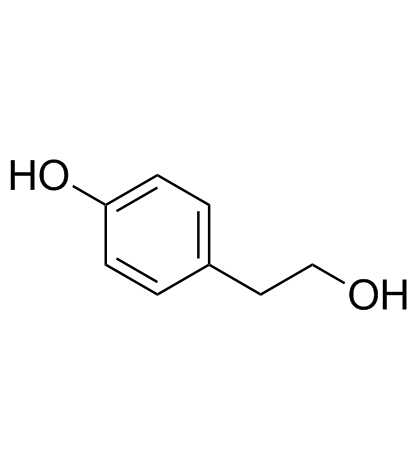 CAS#:501-94-0
CAS#:501-94-0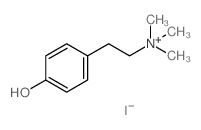 CAS#:1976-98-3
CAS#:1976-98-3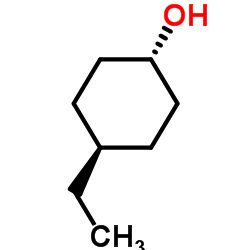 CAS#:19781-62-5
CAS#:19781-62-5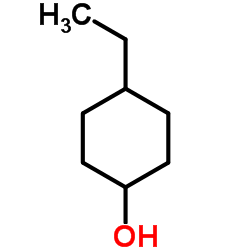 CAS#:19781-61-4
CAS#:19781-61-4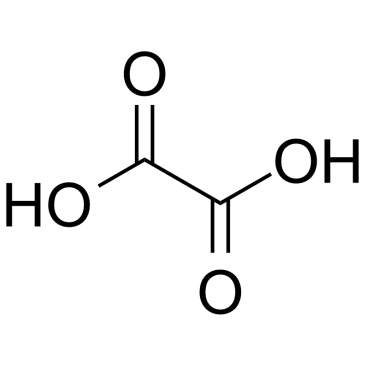 CAS#:144-62-7
CAS#:144-62-7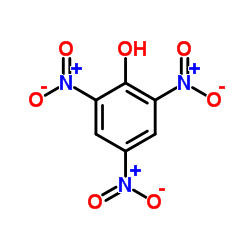 CAS#:88-89-1
CAS#:88-89-1![2,6-dibromo-4-[2-(dimethylamino)ethyl]phenol structure](https://image.chemsrc.com/caspic/449/325151-61-9.png) CAS#:325151-61-9
CAS#:325151-61-9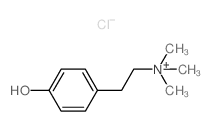 CAS#:3761-58-8
CAS#:3761-58-8
