Betahistine Mesylate
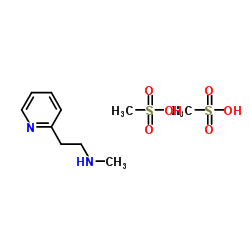
Betahistine Mesylate structure
|
Common Name | Betahistine Mesylate | ||
|---|---|---|---|---|
| CAS Number | 54856-23-4 | Molecular Weight | 328.406 | |
| Density | N/A | Boiling Point | 210.9ºC at 760 mmHg | |
| Molecular Formula | C10H20N2O6S2 | Melting Point | 112°C | |
| MSDS | N/A | Flash Point | 96.7ºC | |
Use of Betahistine MesylateBetahistine mesylate is an orally active histamine H1 receptor agonist and a H3 receptor antagonist[1]. Betahistine mesylate is used for the study of rheumatoid arthritis (RA)[3]. |
| Name | Betahistine Mesylate |
|---|---|
| Synonym | More Synonyms |
| Description | Betahistine mesylate is an orally active histamine H1 receptor agonist and a H3 receptor antagonist[1]. Betahistine mesylate is used for the study of rheumatoid arthritis (RA)[3]. |
|---|---|
| Related Catalog | |
| In Vitro | Betahistine mesylate (0-10 μM) inhibits [125I]iodoproxyfan binding to membranes of CHO (rH3(445)R) and CHO (hH3(445)R) cells with IC50 values of 1.9 μM and 3.3 μM, respectively. Lead to Ki values of 1.4 μM and 2.5 μM, respectively[2]. Betahistine mesylate (0-10 μM) has a regulating function on cAMP formation in CHO (rH3(445)R), CHO (rH3(413)R), and CHO (hH3(445)R) cells. At low concentrations, Betahistine mesylate behaves an apparent inverse agonist, and progressively enhances cAMP formation with EC50 values of 0.1 nM, 0.05 nM and 0.3 nM, respectively. In contrast, at concentrations higher than 10 nM, Betahistine mesylate inhibits cAMP formation with an EC50 value of 0.1 μM in CHO (rH3(445)R) and full agonist activity[2]. |
| In Vivo | Betahistine mesylate (intraperitoneal or oral administration; 0.1-30 mg/kg; single dose) with acute administration has increased tele-methylhistamine (t-MeHA) levels with an ED50 of 0.4 mg/kg, indicating the inverse agonism. Besides, after acute oral administration, it increases t-MeHA levels with an ED50 of 2 mg/kg in male Swissmice[2]. Betahistine mesylate (oral adminstration; 1 and 5 mg/kg; daily for 3 weeks) attenuates the severity of arthritis and reduces the levels of pro-inflammatory cytokines in the paw tissues of CIA mice[3]. Animal Model: Collagen-induced arthritis (CIA) DBA/1 male mouse model[3] Dosage: 1 mg/kg; 5mg/kg Administration: Oral adminstration; day 21 to day 42 after a 21-day CIA induction Result: Ameliorated mouse CIA by decreasing joint destruction. |
| References |
| Boiling Point | 210.9ºC at 760 mmHg |
|---|---|
| Melting Point | 112°C |
| Molecular Formula | C10H20N2O6S2 |
| Molecular Weight | 328.406 |
| Flash Point | 96.7ºC |
| Exact Mass | 328.076263 |
| PSA | 150.42000 |
| LogP | 2.40400 |
| InChIKey | ZBJJDYGJCNTNTH-UHFFFAOYSA-N |
| SMILES | CNCCc1ccccn1.CS(=O)(=O)O.CS(=O)(=O)O |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RTECS | UT2970200 |
|---|---|
| HS Code | 2933399090 |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Comparison of the therapeutic efficacy of a fixed low-dose combination of cinnarizine and dimenhydrinate with betahistine in vestibular neuritis: a randomized, double-blind, non-inferiority study.
Clin. Drug Investig. 32(6) , 387-99, (2012) Vestibular neuritis (VN) is a strongly disabling disease of the peripheral vestibular system. Rapid and effective relief of symptoms is important to allow patients to promptly return to normal physica... |
|
|
Betahistine or Cinnarizine for treatment of Meniere's disease.
Med. Arh. 66(6) , 396-8, (2012) Meniere's disease is a condition with sudden attacks of vertigo with nausea and vomiting accompanied by loss of hearing and buzzing sensation in the ears, most commonly unilateral. The exact cause of ... |
|
|
Vestibular rehabilitation with virtual reality in Ménière's disease.
Braz. J. Otorhinolaryngol. 79(3) , 366-74, (2013) Virtual reality technology can provide a wide range of sensory stimuli to generate conflicts of varying degrees of complexity in a safe environment.To verify the effect of a virtual realitybased balan... |
|
Name: Discovery of Small Molecules to Inhibit Human Cytomegalovirus Nuclear Egress
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: HCMV UL50
External Id: HMS1262
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: MELAS cybrid survival enhancement under low glucose conditions
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: N/A
External Id: HMS1419
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
|
Name: Inhibition of cell viability relative to arbidol control (inhibition index > 1 indica...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303819
|
| 2-Pyridineethanamine, N-methyl-, methanesulfonate (1:2) |
| N-Methyl-2-pyridineethanamine Dimethanesulfonate |
| MFCD00211321 |
| N-Methyl-2-(2-pyridinyl)ethanamine methanesulfonate (1:2) |
| EINECS 259-377-7 |
| N-Methyl-2-(pyridin-2-yl)ethanamine methanesulfonate (1:2) |
| methanesulfonic acid,N-methyl-2-pyridin-2-ylethanamine |