guanethidine
Modify Date: 2025-08-25 00:07:09
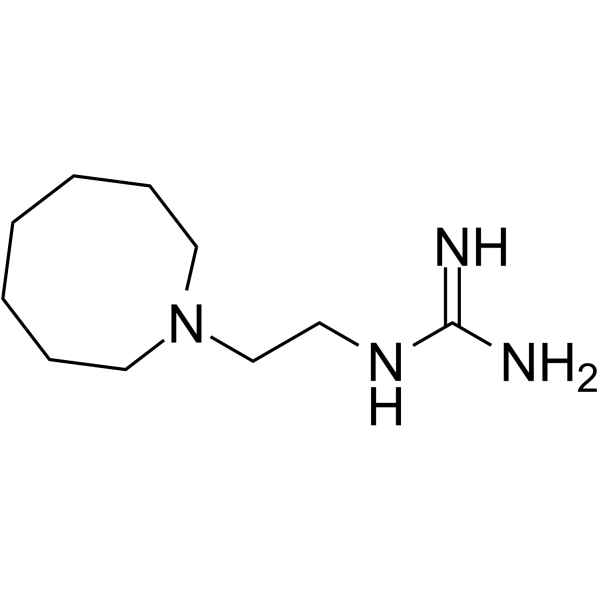
guanethidine structure
|
Common Name | guanethidine | ||
|---|---|---|---|---|
| CAS Number | 55-65-2 | Molecular Weight | 198.30800 | |
| Density | 1.13g/cm3 | Boiling Point | 345.6ºC at 760mmHg | |
| Molecular Formula | C10H22N4 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 162.8ºC | |
Use of guanethidineGuanethidine sulphate was synthesized in 1959. Guanethidine is thought to lowing blood pressure by interfering with the metabolism of chemical transmitter substances in post-ganglionic sympathetic nerve fibres. |
| Name | guanethidine |
|---|---|
| Synonym | More Synonyms |
| Description | Guanethidine sulphate was synthesized in 1959. Guanethidine is thought to lowing blood pressure by interfering with the metabolism of chemical transmitter substances in post-ganglionic sympathetic nerve fibres. |
|---|---|
| In Vitro | Ablation of sympathetic fibers is associated with a loss of rat endothelial cell marker (RECA), but no significant effect of guanethidine was found on the survival of endothelial cells and mesenchymal stem cells in vitro[1]. . |
| In Vivo | Guanethidine (30 mg/kg, s.c., 1 h) unaffected IL-18 hypernociception in TNFR1(-/-) mice as a sympathetic blocker[2]. . Animal Model: Wild-type (WT) Balb/c,TNFR1(-/-)and IFN-γ-γ(-/-) mice[2]. Dosage: 30 mg/kg Administration: Guanethidine (30 mg/kg, s.c., 1 h, diluted in saline) Result: Pre-treatment with guanethidine (sympathetic blocker) unaffected IL-18 hypernociception in TNFR1(-/-) mice. |
| References |
| Density | 1.13g/cm3 |
|---|---|
| Boiling Point | 345.6ºC at 760mmHg |
| Molecular Formula | C10H22N4 |
| Molecular Weight | 198.30800 |
| Flash Point | 162.8ºC |
| Exact Mass | 198.18400 |
| PSA | 65.14000 |
| LogP | 1.86440 |
| Index of Refraction | 1.4910 (estimate) |
| InChIKey | ACGDKVXYNVEAGU-UHFFFAOYSA-N |
| SMILES | NC(N)=NCCN1CCCCCCC1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~% 
guanethidine CAS#:55-65-2 |
| Literature: US4656291 A1, ; |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
|
Name: DRUGMATRIX: Opiate delta1 (OP1, DOP) radioligand binding (ligand: [3H] Naltrindole)
Source: ChEMBL
Target: Delta-type opioid receptor
External Id: CHEMBL1909180
|
|
Name: DRUGMATRIX: Nitric Oxide Synthase, Inducible (iNOS) enzyme inhibition (substrate: L-A...
Source: ChEMBL
Target: Nitric oxide synthase, inducible
External Id: CHEMBL1909179
|
|
Name: DRUGMATRIX: Opiate mu (OP3, MOP) radioligand binding (ligand: [3H] Diprenorphine)
Source: ChEMBL
Target: Mu-type opioid receptor
External Id: CHEMBL1909182
|
|
Name: DRUGMATRIX: Opiate kappa (OP2, KOP) radioligand binding (ligand: [3H] Diprenorphine)
Source: ChEMBL
Target: Kappa-type opioid receptor
External Id: CHEMBL1909181
|
|
Name: DRUGMATRIX: Phosphodiesterase PDE3 enzyme inhibition (substrate: [3H]cAMP + cAMP)
Source: ChEMBL
Target: N/A
External Id: CHEMBL1909184
|
|
Name: DRUGMATRIX: Phorbol Ester radioligand binding (ligand: [3H] PDBu)
Source: ChEMBL
Target: N/A
External Id: CHEMBL1909183
|
|
Name: DRUGMATRIX: Phosphodiesterase PDE5 enzyme inhibition (substrate: [3H]cGMP + cGMP)
Source: ChEMBL
Target: cGMP-specific 3',5'-cyclic phosphodiesterase
External Id: CHEMBL1909186
|
|
Name: DRUGMATRIX: Phosphodiesterase PDE4 enzyme inhibition (substrate: [3H]cAMP + cAMP)
Source: ChEMBL
Target: N/A
External Id: CHEMBL1909185
|
|
Name: DRUGMATRIX: Muscarinic M3 radioligand binding (ligand: [3H] N-Methylscopolamine)
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M3
External Id: CHEMBL1909172
|
|
Name: DRUGMATRIX: Muscarinic M2 radioligand binding (ligand: [3H] N-Methylscopolamine)
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M2
External Id: CHEMBL1909171
|
Total 268, Current Page 1 of 27
1
2
3
4
5
| Guanethidinum |
| Octadine |
| GUANETHIDINE |
| (2-<Octahydroazocinyl-(1)>-ethyl)-guanidin |
| Oktadin |
| [2-(hexahydro-1-(2H)-azocinyl)ethyl]guanidine |
| Isobarin |
| Sanotensin |
| 2-[2-(azocan-1-yl)ethyl]guanidine |
| Oktatenzin |
| (2-azocan-1-yl-ethyl)-guanidine |
| Ismelin |
| Eutensol |
| guanethedine |
| Abapresin |