BHPI
Modify Date: 2025-08-25 08:44:27

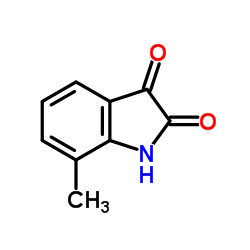
BHPI structure
|
Common Name | BHPI | ||
|---|---|---|---|---|
| CAS Number | 56632-39-4 | Molecular Weight | 331.36500 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C21H17NO3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of BHPIBHPI is a potent inhibitor of nuclear estrogen–ERα-regulated gene expression; elicits sustained ERα-dependent activation of the endoplasmic reticulum (EnR) stress sensor, the unfolded protein response (UPR), and persistent inhibition of protein synthesis.IC50 value:Target: ERα inhibitorBHPI is effective because it elicits sustained ERα-dependent activation of the endoplasmic reticulum (EnR) stress sensor, the unfolded protein response (UPR), and persistent inhibition of protein synthesis. BHPI distorts a newly described action of estrogen-ERα: mild and transient UPR activation. In contrast, BHPI elicits massive and sustained UPR activation, converting the UPR from protective to toxic. In ERα(+) cancer cells, BHPI rapidly hyperactivates plasma membrane PLCγ, generating inositol 1,4,5-triphosphate (IP3), which opens EnR IP3R calcium channels, rapidly depleting EnR Ca(2+) stores. |
| Name | 3,3-bis(4-hydroxyphenyl)-7-methyl-1H-indol-2-one |
|---|---|
| Synonym | More Synonyms |
| Description | BHPI is a potent inhibitor of nuclear estrogen–ERα-regulated gene expression; elicits sustained ERα-dependent activation of the endoplasmic reticulum (EnR) stress sensor, the unfolded protein response (UPR), and persistent inhibition of protein synthesis.IC50 value:Target: ERα inhibitorBHPI is effective because it elicits sustained ERα-dependent activation of the endoplasmic reticulum (EnR) stress sensor, the unfolded protein response (UPR), and persistent inhibition of protein synthesis. BHPI distorts a newly described action of estrogen-ERα: mild and transient UPR activation. In contrast, BHPI elicits massive and sustained UPR activation, converting the UPR from protective to toxic. In ERα(+) cancer cells, BHPI rapidly hyperactivates plasma membrane PLCγ, generating inositol 1,4,5-triphosphate (IP3), which opens EnR IP3R calcium channels, rapidly depleting EnR Ca(2+) stores. |
|---|---|
| Related Catalog | |
| References |
| Molecular Formula | C21H17NO3 |
|---|---|
| Molecular Weight | 331.36500 |
| Exact Mass | 331.12100 |
| PSA | 73.05000 |
| LogP | 3.77780 |
| Storage condition | 2-8℃ |
|
~% 
BHPI CAS#:56632-39-4 |
| Literature: Uddin, Muhammed K.; Reignier, Serge G.; Coulter, Tom; Montalbetti, Christian; Granaes, Charlotta; Butcher, Steven; Krog-Jensen, Christian; Felding, Jakob Bioorganic and Medicinal Chemistry Letters, 2007 , vol. 17, # 10 p. 2854 - 2857 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
| HMS2153B10 |
| 7-methyl-oxyphenisatine |
| BHPI |