Dipyridamole
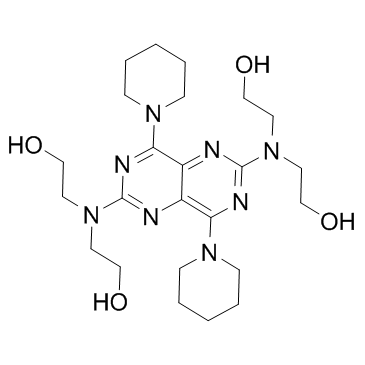
Dipyridamole structure
|
Common Name | Dipyridamole | ||
|---|---|---|---|---|
| CAS Number | 58-32-2 | Molecular Weight | 504.626 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 806.5±75.0 °C at 760 mmHg | |
| Molecular Formula | C24H40N8O4 | Melting Point | 165-166ºC | |
| MSDS | Chinese USA | Flash Point | 441.5±37.1 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of DipyridamoleDipyridamole (Persantine) is a phosphodiesterase inhibitor that blocks uptake and metabolism of adenosine by erythrocytes and vascular endothelial cells.Target: Phosphodiesterase (PDE)Dipyridamole concentrations of 1 nmol/ml blood caused 90% inhibition of adenosine metabolism. Dipyridamole at therapeutic concentrations causes significant inhibition of adenosine metabolism in whole blood [1]. Dipyridamole has a dose-dependent inhibitory effect on thromboxane synthesis which was independent of aggregation. Dipyridamole also inhibited malonyldialdehyde production in response to both thrombin and arachidonic acid [2]. Dipyridamole enhances platelet inhibition by amplifying the signaling of the NO donor sodium nitroprusside. These data support the concept that enhancement of endothelium-dependent NO/cGMP-mediated signaling may be an important in vivo component of dipyridamole action [3]. |
| Name | dipyridamole |
|---|---|
| Synonym | More Synonyms |
| Description | Dipyridamole (Persantine) is a phosphodiesterase inhibitor that blocks uptake and metabolism of adenosine by erythrocytes and vascular endothelial cells.Target: Phosphodiesterase (PDE)Dipyridamole concentrations of 1 nmol/ml blood caused 90% inhibition of adenosine metabolism. Dipyridamole at therapeutic concentrations causes significant inhibition of adenosine metabolism in whole blood [1]. Dipyridamole has a dose-dependent inhibitory effect on thromboxane synthesis which was independent of aggregation. Dipyridamole also inhibited malonyldialdehyde production in response to both thrombin and arachidonic acid [2]. Dipyridamole enhances platelet inhibition by amplifying the signaling of the NO donor sodium nitroprusside. These data support the concept that enhancement of endothelium-dependent NO/cGMP-mediated signaling may be an important in vivo component of dipyridamole action [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 806.5±75.0 °C at 760 mmHg |
| Melting Point | 165-166ºC |
| Molecular Formula | C24H40N8O4 |
| Molecular Weight | 504.626 |
| Flash Point | 441.5±37.1 °C |
| Exact Mass | 504.317261 |
| PSA | 145.44000 |
| LogP | -1.22 |
| Vapour Pressure | 0.0±3.0 mmHg at 25°C |
| Index of Refraction | 1.670 |
| InChIKey | IZEKFCXSFNUWAM-UHFFFAOYSA-N |
| SMILES | OCCN(CCO)c1nc(N2CCCCC2)c2nc(N(CCO)CCO)nc(N3CCCCC3)c2n1 |
| Storage condition | −20°C |
| Water Solubility | DMSO: soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | KK7450000 |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Hypoxanthine uptake by skeletal muscle microvascular endothelial cells from equilibrative nucleoside transporter 1 (ENT1)-null mice: effect of oxidative stress.
Microvasc. Res. 98 , 16-22, (2015) Adenosine is an endogenous regulator of vascular tone. This activity of adenosine is terminated by its uptake and metabolism by microvascular endothelial cells (MVEC). The predominant transporter invo... |
|
|
Oxidative stress modulates nucleobase transport in microvascular endothelial cells
Microvasc. Res. 95 , 68-75, (2014) Purine nucleosides and nucleobases play key roles in the physiological response to vascular ischemia/reperfusion events. The intra- and extracellular concentrations of these compounds are controlled, ... |
|
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
| 2,2',2'',2'''-[(4,8-Dipiperidin-1-ylpyrimido[5,4-d]pyrimidin-2,6-diyl)dinitrilo]tetraethanol |
| RA 8 |
| Piroan |
| EINECS 200-374-7 |
| Dipyridamole |
| Coridil |
| Dypyridamole |
| Apricor |
| Anginal |
| Natyl |
| 2,2',2'',2'''-{[4,8-Di(piperidin-1-yl)pyrimido[5,4-d]pyrimidine-2,6-diyl]dinitrilo}tetraethanol |
| 2,2',2'',2'''-{[4,8-Di(1-piperidinyl)pyrimido[5,4-d]pyrimidine-2,6-diyl]dinitrilo}tetraethanol |
| Corosan |
| Coroxin |
| MFCD00010555 |
| Coribon |
![2,6-DICHLORO-4,8-DIPIPERIDINOPYRIMIDINO[5,4-D]PYRIMIDINE Structure](https://image.chemsrc.com/caspic/334/7139-02-8.png) CAS#:7139-02-8
CAS#:7139-02-8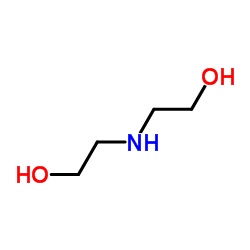 CAS#:111-42-2
CAS#:111-42-2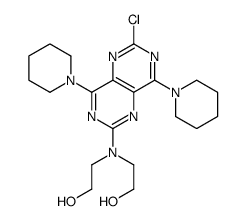 CAS#:54093-92-4
CAS#:54093-92-4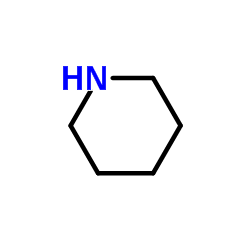 CAS#:110-89-4
CAS#:110-89-4 CAS#:32980-71-5
CAS#:32980-71-5
