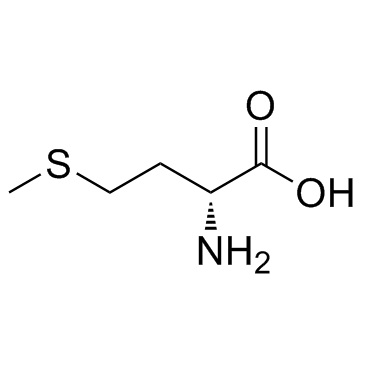
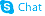L-Homoserine
L-Homoserine structure
|
Common Name | L-Homoserine | ||
|---|---|---|---|---|
| CAS Number | 6027-21-0 | Molecular Weight | 119.12 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 368.7±32.0 °C at 760 mmHg | |
| Molecular Formula | C4H9NO3 | Melting Point | 205ºC | |
| MSDS | Chinese USA | Flash Point | 176.8±25.1 °C | |
Use of L-HomoserineH-D-Homoser-OH is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | D-homoserine |
|---|---|
| Synonym | More Synonyms |
| Description | H-D-Homoser-OH is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 368.7±32.0 °C at 760 mmHg |
| Melting Point | 205ºC |
| Molecular Formula | C4H9NO3 |
| Molecular Weight | 119.12 |
| Flash Point | 176.8±25.1 °C |
| Exact Mass | 119.058243 |
| PSA | 83.55000 |
| LogP | -1.13 |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C |
| Index of Refraction | 1.511 |
| Storage condition | 2~8°C |
|
~99% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Jamieson, Andrew G.; Boutard, Nicolas; Beauregard, Kim; Bodas, Mandar S.; Ong, Huy; et al. Journal of the American Chemical Society, 2009 , vol. 131, p. 7917 - 7927 |
|
~30% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Koriyama, Yuji; Nozawa, Akihiro; Hayakawa, Ryuuichirou; Shimizu, Makoto Tetrahedron, 2002 , vol. 58, # 47 p. 9621 - 9628 |
|
~81% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Shiraiwa, Tadashi; Miyazaki, Hideya; Ohta, Atsushi; Waki, Yukitaka; Yasuda, Masahiro; Morishita, Toshimitsu; Kurokawa, Hidemoto Chemical and Pharmaceutical Bulletin, 1996 , vol. 44, # 12 p. 2322 - 2325 |
|
~% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Tetrahedron, , vol. 44, # 2 p. 637 - 642 |
|
~% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Archives of Biochemistry, , vol. 42, p. 212,214 |
|
~% 
L-Homoserine CAS#:6027-21-0 |
| Literature: Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences, , vol. 59, # 4 p. 451 - 467 |
| HS Code | 2922509090 |
|---|---|
| Summary | 2922509090. other amino-alcohol-phenols, amino-acid-phenols and other amino-compounds with oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
A Simple Enzymatic Method for Production of a Wide Variety of D-Amino Acids Using L-Amino Acid Oxidase from Rhodococcus sp. AIU Z-35-1.
Enzyme Res. 2010 , 567210., (2010) A simple enzymatic method for production of a wide variety of D-amino acids was developed by kinetic resolution of DL-amino acids using L-amino acid oxidase (L-AAO) with broad substrate specificity fr... |
|
|
Engineering d-amino acid containing novel protease inhibitors using catalytic site architecture.
Bioorg. Med. Chem. 14 , 214-236, (2006) The mechanism of proteolysis by serine proteases is a reasonably well-understood process. Typically, a histidine residue acting as a general base deprotonates the catalytic serine residue and the hydr... |
|
|
Structure of the O-antigen of Acinetobacter lwoffii EK30A; identification of d-homoserine, a novel non-sugar component of bacterial polysaccharides.
Org. Biomol. Chem. 8 , 3571-3577, (2010) We established a peculiar structure of the O-specific polysaccharide (O-antigen) of a psychrotrophic strain of Acinetobacter lwoffii, EK30A, isolated from a 1.6-1.8 million-year-old Siberian permafros... |
| Homoserine |
| (2R)-2-amino-4-hydroxybutanoic acid |
| (2S)-2-Amino-4-hydroxybutyric Acid |
| (S)-2-amino-4-hydroxy-Butanoic acid |
| (R)-homoserine |
| (S)-Homoserine |
| (R)-3-amino-5-hydroxypentanoic acid |
| (R)-(+)-2-Amino-4-hydroxybutyric acid |
| Homoserine,D |
| MFCD00077786 |
| AmbotzHAA1028 |
| H-D-HoSer-OH |
| R-2-Amino-4-hydroxybutyric acid (D-Homoser) |
| D-Homoserine |
| (S)-2-Amino-4-hydroxybutanoic acid |

![N-[(Benzyloxy)carbonyl]-D-homoserine structure](https://www.chemsrc.com/caspic/475/41088-85-1.png) CAS#:41088-85-1
CAS#:41088-85-1