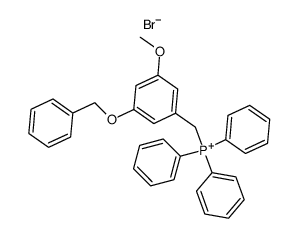
Batatasin IV
Modify Date: 2025-08-25 18:18:40
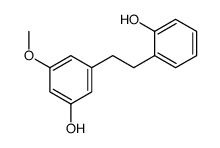
Batatasin IV structure
|
Common Name | Batatasin IV | ||
|---|---|---|---|---|
| CAS Number | 60347-67-3 | Molecular Weight | 244.29 | |
| Density | 1.198g/cm3 | Boiling Point | 416.5ºC at 760 mmHg | |
| Molecular Formula | C15H16O3 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 205.7ºC | |
Use of Batatasin IVBatatasin IV is an LTA4H inhibitor with anti-inflammatory activity. Batatasin IV is also a natural product that can be obtained from Dioscorea batatus. Batatasin IV can be used in research in the area of inflammation[1]. |
| Name | 3-[2-(2-Hydroxyphenyl)ethyl]-5-methoxyphenol |
|---|---|
| Synonym | More Synonyms |
| Description | Batatasin IV is an LTA4H inhibitor with anti-inflammatory activity. Batatasin IV is also a natural product that can be obtained from Dioscorea batatus. Batatasin IV can be used in research in the area of inflammation[1]. |
|---|---|
| Related Catalog | |
| Target |
LTA4H[1]. |
| References |
| Density | 1.198g/cm3 |
|---|---|
| Boiling Point | 416.5ºC at 760 mmHg |
| Molecular Formula | C15H16O3 |
| Molecular Weight | 244.29 |
| Flash Point | 205.7ºC |
| Exact Mass | 244.11000 |
| PSA | 49.69000 |
| LogP | 2.89160 |
| Index of Refraction | 1.61 |
| InChIKey | IUMFLNFLJUUODE-UHFFFAOYSA-N |
| SMILES | COc1cc(O)cc(CCc2ccccc2O)c1 |
| Storage condition | 2-8°C |
| HS Code | 2909500000 |
|---|
|
~% 
Batatasin IV CAS#:60347-67-3 |
| Literature: Hashimoto,T.; Tajima,M. Phytochemistry (Elsevier), 1978 , vol. 17, p. 1179 - 1184 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
| HS Code | 2909500000 |
|---|---|
| Summary | 2909500000 ether-phenols, ether-alcohol-phenols and their halogenated, sulphonated, nitrated or nitrosated derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:5.5% General tariff:30.0% |
|
Name: Antifungal activity against Candida albicans by colorimetric method
Source: ChEMBL
Target: Candida albicans
External Id: CHEMBL914257
|
|
Name: Inhibition of COX1 by radioimmunoassay method
Source: ChEMBL
Target: Prostaglandin G/H synthase 1
External Id: CHEMBL914258
|
|
Name: Inhibition of COX2 by radioimmunoassay method
Source: ChEMBL
Target: Prostaglandin G/H synthase 2
External Id: CHEMBL914259
|
|
Name: Induction of NGF production in rat C6 cells at 20 uM after 24 hrs by ELISA relative t...
Source: ChEMBL
Target: C6
External Id: CHEMBL1762878
|
|
Name: Cytotoxicity against KB cells by colorimetric method
Source: ChEMBL
Target: KB
External Id: CHEMBL914260
|
|
Name: Cytotoxicity against rat C6 cells assessed as viable cells at 20 uM after 24 hrs by M...
Source: ChEMBL
Target: C6
External Id: CHEMBL1762879
|
|
Name: Antimycobacterial activity against Mycobacterium tuberculosis H37Ra by microplate Ala...
Source: ChEMBL
Target: Mycobacterium tuberculosis
External Id: CHEMBL914255
|
|
Name: Antimalarial activity against multidrug-resistant Plasmodium falciparum K1 by micro c...
Source: ChEMBL
Target: Plasmodium falciparum
External Id: CHEMBL914256
|
|
Name: Inhibition of ovine COX2 by colorimetric assay
Source: ChEMBL
Target: Prostaglandin G/H synthase 2
External Id: CHEMBL1018881
|
|
Name: Antioxidant activity assessed as superoxide radical scavenging activity after 20 mins...
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL1018879
|
Total 13, Current Page 1 of 2
1
2
| Batatasin IV <2',3-Dihydroxy-5-methoxybibenzyl> |
| 2',3-Dihydroxy-5-methoxybibenzyl |
| 3-[2-(2-hydroxyphenyl)ethyl]-5-methoxy-phenol |
| 3,2'-dihydroxy-5-methoxybibenzyl |
| Batatasin IV |