Homopterocarpin
Modify Date: 2025-08-24 09:52:54

Homopterocarpin structure
|
Common Name | Homopterocarpin | ||
|---|---|---|---|---|
| CAS Number | 606-91-7 | Molecular Weight | 284.306 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 395.0±42.0 °C at 760 mmHg | |
| Molecular Formula | C17H16O4 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 140.4±34.7 °C | |
Use of HomopterocarpinHomopterocarpin is an isoflavonoid that can be isolated from Pterocarpus erinaceus. Homopterocarpin has hepatoprotective and antioxidant properties. Homopterocarpin is a competitive reversible inhibitor of human monoamine oxidase-B with an IC50 and a Ki of 0.72 and 0.21 μM for hMAO-B, respectively. Homopterocarpin can be used for the research of liver injury and oxidative stress[1][2]. |
| Name | Homopterocarpin |
|---|---|
| Synonym | More Synonyms |
| Description | Homopterocarpin is an isoflavonoid that can be isolated from Pterocarpus erinaceus. Homopterocarpin has hepatoprotective and antioxidant properties. Homopterocarpin is a competitive reversible inhibitor of human monoamine oxidase-B with an IC50 and a Ki of 0.72 and 0.21 μM for hMAO-B, respectively. Homopterocarpin can be used for the research of liver injury and oxidative stress[1][2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 395.0±42.0 °C at 760 mmHg |
| Molecular Formula | C17H16O4 |
| Molecular Weight | 284.306 |
| Flash Point | 140.4±34.7 °C |
| Exact Mass | 284.104858 |
| PSA | 36.92000 |
| LogP | 3.14 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.589 |
| InChIKey | VPGIGLKLCFOWDN-YOEHRIQHSA-N |
| SMILES | COc1ccc2c(c1)OC1c3ccc(OC)cc3OCC21 |
| Hazard Codes | Xi |
|---|---|
| HS Code | 2932999099 |
| Precursor 0 | |
|---|---|
| DownStream 2 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Cytotoxicity against human HL60 cells after 72 hrs by MTT assay
Source: ChEMBL
Target: HL-60
External Id: CHEMBL979286
|
|
Name: Cytotoxicity against human CEM cells after 72 hrs by MTT assay
Source: ChEMBL
Target: CCRF-CEM
External Id: CHEMBL979285
|
|
Name: Cytotoxicity against human MCF7 cells after 72 hrs by MTT assay
Source: ChEMBL
Target: MCF7
External Id: CHEMBL979288
|
|
Name: Cytotoxicity against human HCT8 cells after 72 hrs by MTT assay
Source: ChEMBL
Target: HCT-8
External Id: CHEMBL979287
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Screen for inhibitors of RMI FANCM (MM2) intereaction
Source: 11908
Target: N/A
External Id: RMI-FANCM-MM2
|
|
Name: Primary high throughput screening by co-culture imaging for identification hits as a ...
Source: 23209
Target: N/A
External Id: UIHTS20180925
|
|
Name: Spectrum HTS for Inhibitors of Aerobactin Synthetase IucA
Source: 23265
Target: IucA Synthetase from hypervirulent Klebsiella pneumoniae hvKP1
External Id: IucA Pilot Assay Spectrum Library
|
|
Name: Inhibition of sodium fluorescein uptake in OATP1B3-transfected CHO cells at an equimo...
Source: ChEMBL
Target: Solute carrier organic anion transporter family member 1B3
External Id: CHEMBL3039491
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
Total 18, Current Page 1 of 2
1
2
| (6aR,11aR)-3,9-Dimethoxy-6a,11a-dihydro-6H-[1]benzofuro[3,2-c]chromene |
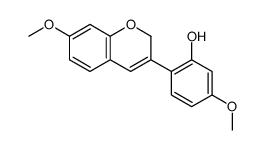 CAS#:3187-50-6
CAS#:3187-50-6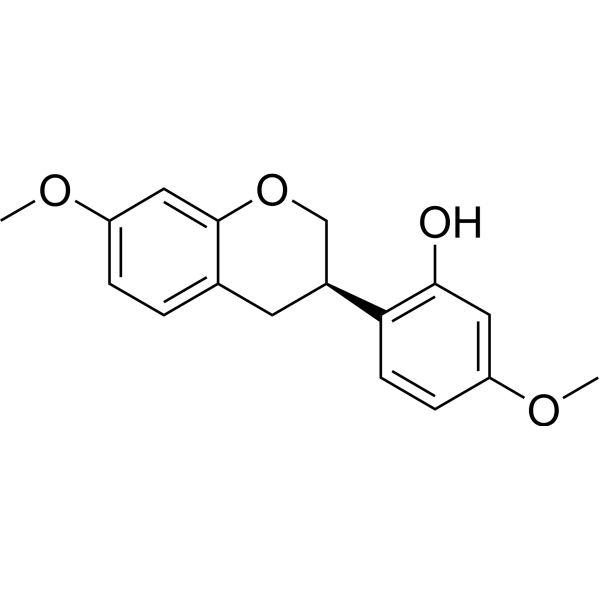 CAS#:60102-29-6
CAS#:60102-29-6