5-Methoxy-3-indoleaceate
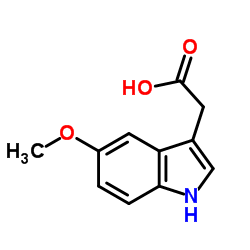
5-Methoxy-3-indoleaceate structure
|
Common Name | 5-Methoxy-3-indoleaceate | ||
|---|---|---|---|---|
| CAS Number | 608-07-1 | Molecular Weight | 205.210 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 445.9±30.0 °C at 760 mmHg | |
| Molecular Formula | C11H14N2O | Melting Point | 245-250 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 223.5±24.6 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | O-methylserotonin |
|---|---|
| Synonym | More Synonyms |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 445.9±30.0 °C at 760 mmHg |
| Melting Point | 245-250 °C (dec.)(lit.) |
| Molecular Formula | C11H14N2O |
| Molecular Weight | 205.210 |
| Flash Point | 223.5±24.6 °C |
| Exact Mass | 205.073898 |
| PSA | 51.04000 |
| LogP | 1.34 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.656 |
| InChIKey | JTEJPPKMYBDEMY-UHFFFAOYSA-N |
| SMILES | COc1ccc2[nH]cc(CCN)c2c1 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NL4110000 |
| HS Code | 2933990090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro.
Mol. Cell. Endocrinol. 404 , 1-8, (2015) Melatonin and its metabolites including 6-hydroxymelatonin (6(OH)M), N(1)-acetyl-N(2)-formyl-5-methoxykynuramine (AFMK) and 5-methoxytryptamine (5MT) are endogenously produced in human epidermis. This... |
|
|
The antioxidant behaviour of melatonin and structural analogues during lipid peroxidation depends not only on their functional groups but also on the assay system.
Biochem. Biophys. Res. Commun. 423(4) , 873-7, (2012) There is no general agreement yet on the antioxidant effect of pineal indoles against lipid peroxidation. Accordingly, the main goal of the present work was to study the antioxidant activity of melato... |
|
|
Hippocampal serotonin depletion is related to the presence of generalized tonic-clonic seizures, but not to psychiatric disorders in patients with temporal lobe epilepsy.
Epilepsy Res. 111 , 18-25, (2015) Previous studies suggest that concentration of serotonin ([5-HT]) plays a pathogenic role in various types of epilepsy inhibiting seizures. However, most have not considered the clinical variables of ... |
|
Name: Evaluated for the binding affinity to hippocampus striatal membranes at 5-hydroxytryp...
Source: ChEMBL
Target: 5-hydroxytryptamine receptor 1A
External Id: CHEMBL615963
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Antibacterial activity against Staphylococcus aureus MRSA ATCC 43300 (CO-ADD:GP_020);...
Source: ChEMBL
Target: Staphylococcus aureus
External Id: CHEMBL4296184
|
|
Name: Antibacterial activity against Klebsiella pneumoniae MDR ATCC 70063 (CO-ADD:GN_003); ...
Source: ChEMBL
Target: Klebsiella pneumoniae
External Id: CHEMBL4296186
|
|
Name: Binding affinity at rat 5-hydroxytryptamine 7 receptor.
Source: ChEMBL
Target: 5-hydroxytryptamine receptor 7
External Id: CHEMBL620766
|
|
Name: Antibacterial activity against Escherichia coli ATCC 25922 (CO-ADD:GN_001); MIC in CA...
Source: ChEMBL
Target: Escherichia coli
External Id: CHEMBL4296185
|
|
Name: Agonistic activity against rat 5-HT7 adenyl cyclase expressed in HEK293 cells; Active
Source: ChEMBL
Target: 5-hydroxytryptamine receptor 7
External Id: CHEMBL619703
|
|
Name: Primary Screen Inhibitors of CD40 Signaling in BL2 Cells Measured in Cell-Based Syste...
Source: Broad Institute
Target: N/A
External Id: 7124-01_Inhibitor_SinglePoint_HTS_Activity
|
| 5-Methoxy-3-indoleacetic acid |
| Mexamine base |
| (5-Methoxy-1H-indol-3-yl)acetic acid |
| O-METHYLSEROTONIN |
| 5MOT |
| 5-Methoxyindoleacetic acid |
| EINECS 200-637-6 |
| 5-Methoxytryptamine |
| MFCD00005662 |
| 5-Methoxy-3-indoleacetate |
| 5-Methoxy-3-indoleaceate |
| 5-methoxy-1H-3-indoleacetic acid |
| 5-Methoxyindole-3-acetic acid |
| Meksamin |
| Methoxyindoleacetic acid |
| O-methyl-5-HT |
| MEXAMINE |
 CAS#:61675-19-2
CAS#:61675-19-2 CAS#:73-31-4
CAS#:73-31-4 CAS#:68935-49-9
CAS#:68935-49-9 CAS#:1006-94-6
CAS#:1006-94-6 CAS#:67199-10-4
CAS#:67199-10-4 CAS#:102016-76-2
CAS#:102016-76-2 CAS#:627086-10-6
CAS#:627086-10-6![methyl N-[2-(1H-indol-3-yl)ethyl]carbamate Structure](https://image.chemsrc.com/caspic/015/58635-45-3.png) CAS#:58635-45-3
CAS#:58635-45-3![N-[2-(5-methoxy-1H-indol-3-yl)ethyl]-3-oxobutanamide structure](https://image.chemsrc.com/caspic/329/112081-40-0.png) CAS#:112081-40-0
CAS#:112081-40-0![1-[2-(5-methoxy-1H-indol-3-yl)ethyl]-3-prop-2-enylthiourea structure](https://image.chemsrc.com/caspic/365/111915-67-4.png) CAS#:111915-67-4
CAS#:111915-67-4![N-[2-(5-methoxy-1H-indol-3-yl)ethyl]formamide structure](https://image.chemsrc.com/caspic/015/104510-12-5.png) CAS#:104510-12-5
CAS#:104510-12-5 CAS#:50-67-9
CAS#:50-67-9 CAS#:3589-73-9
CAS#:3589-73-9 CAS#:2009-03-2
CAS#:2009-03-2![N-[2-(5-methoxy-1H-indol-3-yl)ethyl]pyridine-3-carboxamide structure](https://image.chemsrc.com/caspic/201/29745-42-4.png) CAS#:29745-42-4
CAS#:29745-42-4 CAS#:96096-55-8
CAS#:96096-55-8![N-[2-(5-methoxyindol-3-yl)ethyl]-cyclopropylcarboxamide structure](https://image.chemsrc.com/caspic/238/139564-01-5.png) CAS#:139564-01-5
CAS#:139564-01-5
