Dopamine hydrochloride
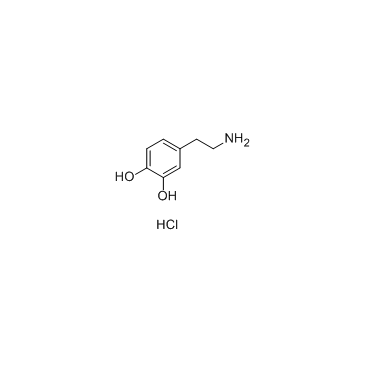
Dopamine hydrochloride structure
|
Common Name | Dopamine hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 62-31-7 | Molecular Weight | 189.639 | |
| Density | N/A | Boiling Point | 337.7ºC at 760 mmHg | |
| Molecular Formula | C8H12ClNO2 | Melting Point | 248-250 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 51.8 °F | |
| Symbol |


GHS07, GHS09 |
Signal Word | Warning | |
Use of Dopamine hydrochlorideDopamine HCl is a catecholamine neurotransmitter present in a wide variety of animals,And a dopamine D1-5 receptors agonist.Target: Dopamine ReceptorDopamine (or 3,4-dihydroxyphenethylamine) is a neuroendocrine transmitter in the catecholamine and phenethylamine families that plays a number of important roles in the brain and bodies of humans. Several important diseases of the nervous system are associated with dysfunctions of the dopamine system. Outside the nervous system, dopamine functions in several parts of the body as a local chemical messenger. In the blood vessels, it inhibits norepinephrine release and acts as a vasodilator; in the kidneys, it increases sodium excretion and urine output; in the pancreas, it reduces insulin production; in the digestive system, it reduces gastrointestinal motility and protects intestinal mucosa; and in the immune system, it reduces the activity of lymphocytes. A variety of important drugs work by altering the way the body makes or uses dopamine. Dopamine itself is available for intravenous injection: although it cannot reach the brain from the bloodstream, its peripheral effects make it useful in the treatment of heart failure or shock, especially in newborn babies. L-DOPA, the metabolic precursor of dopamine, does reach the brain and is the most widely used treatment for Parkinson's disease. From Wikipedia. |
| Name | 3-Hydroxytyramine hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Dopamine HCl is a catecholamine neurotransmitter present in a wide variety of animals,And a dopamine D1-5 receptors agonist.Target: Dopamine ReceptorDopamine (or 3,4-dihydroxyphenethylamine) is a neuroendocrine transmitter in the catecholamine and phenethylamine families that plays a number of important roles in the brain and bodies of humans. Several important diseases of the nervous system are associated with dysfunctions of the dopamine system. Outside the nervous system, dopamine functions in several parts of the body as a local chemical messenger. In the blood vessels, it inhibits norepinephrine release and acts as a vasodilator; in the kidneys, it increases sodium excretion and urine output; in the pancreas, it reduces insulin production; in the digestive system, it reduces gastrointestinal motility and protects intestinal mucosa; and in the immune system, it reduces the activity of lymphocytes. A variety of important drugs work by altering the way the body makes or uses dopamine. Dopamine itself is available for intravenous injection: although it cannot reach the brain from the bloodstream, its peripheral effects make it useful in the treatment of heart failure or shock, especially in newborn babies. L-DOPA, the metabolic precursor of dopamine, does reach the brain and is the most widely used treatment for Parkinson's disease. From Wikipedia. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
| Boiling Point | 337.7ºC at 760 mmHg |
|---|---|
| Melting Point | 248-250 °C(lit.) |
| Molecular Formula | C8H12ClNO2 |
| Molecular Weight | 189.639 |
| Exact Mass | 189.055649 |
| PSA | 66.48000 |
| LogP | 2.10130 |
| InChIKey | CTENFNNZBMHDDG-UHFFFAOYSA-N |
| SMILES | Cl.NCCc1ccc(O)c(O)c1 |
| Stability | Stable. Incompatible with strong oxidizing agents. Combustible. |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS07, GHS09 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H410 |
| Precautionary Statements | P273-P501 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | UN 3077 9/PG 3 |
| WGK Germany | 2 |
| RTECS | UX1092000 |
| HS Code | 29222900 |
| Flash Point(F) | 51.8 °F |
| Flash Point(C) | 11 °C |
|
~96% 
Dopamine hydroc... CAS#:62-31-7 |
| Literature: Kohno; Sasao; Murahashi Bulletin of the Chemical Society of Japan, 1990 , vol. 63, # 4 p. 1252 - 1254 |
|
~76% 
Dopamine hydroc... CAS#:62-31-7 |
| Literature: Velcicky, Juraj; Soicke, Arne; Steiner, Roland; Schmalz, Hans-Guenther Journal of the American Chemical Society, 2011 , vol. 133, # 18 p. 6948 - 6951 |
|
~99% 
Dopamine hydroc... CAS#:62-31-7 |
| Literature: Bernini, Roberta; Crisante, Fernanda; Barontini, Maurizio; Fabrizi, Giancarlo Synthesis, 2009 , # 22 art. no. P09409SS, p. 3838 - 3842 |
|
~% 
Dopamine hydroc... CAS#:62-31-7 |
| Literature: Journal of the American Chemical Society, , vol. 133, # 18 p. 6948 - 6951 |
| Precursor 4 | |
|---|---|
| DownStream 10 | |
| HS Code | 29222900 |
|---|
|
'Dopamine-first' mechanism enables the rational engineering of the norcoclaurine synthase aldehyde activity profile.
FEBS J. 282(6) , 1137-51, (2015) Norcoclaurine synthase (NCS) (EC 4.2.1.78) catalyzes the Pictet-Spengler condensation of dopamine and an aldehyde, forming a substituted (S)-tetrahydroisoquinoline, a pharmaceutically important moiety... |
|
|
Permeation of Dopamine Sulfate through the Blood-Brain Barrier.
PLoS ONE 10 , e0133904, (2015) Dopamine sulfate (DA-3- and DA-4-S) have been determined in the human brain, but it is unclear whether they are locally formed in the central nervous system (CNS), or transported into the CNS from per... |
|
|
Increasing the Collision Rate of Particle Impact Electroanalysis with Magnetically Guided Pt-Decorated Iron Oxide Nanoparticles.
ACS Nano 9 , 7583-95, (2015) An integrated microfluidic/magnetophoretic methodology was developed for improving signal response time and detection limits for the chronoamperometric observation of discrete nanoparticle/electrode i... |
| 3,4-Dihydroxy-b-phenethylamine Hydrochloride |
| 4-(2-aminoéthyl)benzène-1,2-diol chlorhydrate |
| b-(3,4-Dihydroxyphenyl)ethylamine Hydrochloride |
| Tensamin |
| 4-(2-Aminoethyl)benzene-1,2-diol hydrochloride (1:1) |
| Rascordin |
| Pyrocatechol, 4- (2-aminoethyl)-, hydrochloride |
| 1,2-Benzenediol, 4-(2-aminoethyl)-, hydrochloride (1:1) |
| 4-(2-aminoethyl)benzene-1,2-diol,hydrochloride |
| 1,2-Benzenediol, 4- (2-aminoethyl)-, hydrochloride |
| 4-(2-Aminoethyl)-1,2-benzenediol hydrochloride (1:1) |
| 4-(2-aminoethyl)-Pyrocatechol hydrochloride |
| 4-(2-Aminoethyl)benzol-1,2-diolhydrochlorid |
| EINECS 200-527-8 |
| MFCD00012898 |
| 4-(2-Aminoethyl)catechol, hydrochloride |
| β-(3,4-Dihydroxyphenyl)ethylamine hydrochloride |
| Dopamine (hydrochloride) |
![tert-butyl N-[2-(3,4-dihydroxyphenyl)ethyl]carbamate structure](https://image.chemsrc.com/caspic/285/37034-31-4.png)

![N-[2-(3,4-dihydroxyphenyl)ethyl]octadeca-9,12,15-trienamide structure](https://image.chemsrc.com/caspic/238/105955-13-3.png) CAS#:105955-13-3
CAS#:105955-13-3 CAS#:105955-12-2
CAS#:105955-12-2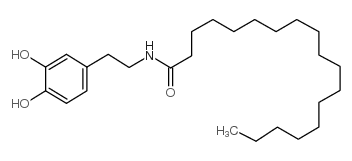 CAS#:105955-10-0
CAS#:105955-10-0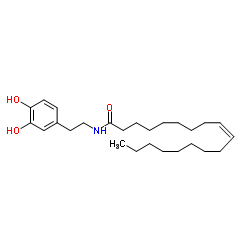 CAS#:105955-11-1
CAS#:105955-11-1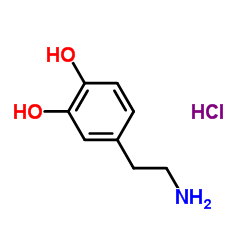 CAS#:51-61-6
CAS#:51-61-6![N-[2-(3,4-Dihydroxyphenyl)ethyl]-2-propenamide structure](https://image.chemsrc.com/caspic/086/201610-44-8.png) CAS#:201610-44-8
CAS#:201610-44-8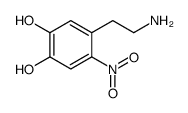 CAS#:21581-49-7
CAS#:21581-49-7 CAS#:2494-12-4
CAS#:2494-12-4 CAS#:10597-60-1
CAS#:10597-60-1 CAS#:102-32-9
CAS#:102-32-9
