sodium gualenate
Modify Date: 2025-08-20 08:36:26

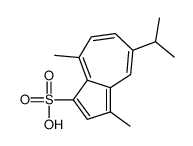
sodium gualenate structure
|
Common Name | sodium gualenate | ||
|---|---|---|---|---|
| CAS Number | 6223-35-4 | Molecular Weight | 300.348 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C15H17NaO3S | Melting Point | 98°C(lit.) | |
| MSDS | N/A | Flash Point | N/A | |
Use of sodium gualenateSodium gualenate (Guaiazulenesulfonate sodium) is a hydrophilic derivative of guaiazulene with excellent anti-inflammatory and wound-healing effects mainly used for the treatment of duodenal ulcer, gastric ulcer and gastritis. |
| Name | sodium,3,8-dimethyl-5-propan-2-ylazulene-1-sulfonate |
|---|---|
| Synonym | More Synonyms |
| Description | Sodium gualenate (Guaiazulenesulfonate sodium) is a hydrophilic derivative of guaiazulene with excellent anti-inflammatory and wound-healing effects mainly used for the treatment of duodenal ulcer, gastric ulcer and gastritis. |
|---|---|
| Related Catalog | |
| In Vitro | Sodium gualenate is an unstable compound, which is gradually decomposed in the solid state at room temperature. When heated, Sodium gualenate decomposes almost completely within 1 week. It was found that a kneaded mixture of Sodium gualenate and cornstarch (weight ratio; 1:250) for tableting with water is stable. So, during production, Sodium gualenate could be stabilized using water[1]. Sodium gualenate slightly inhibits the histamine release from rat peritoneal mast cells and strongly inhibits the leukocyte emigration induced by fMLP[2]. |
| In Vivo | Sodium gualenate has been frequently used for the treatment of human gastritis. Cytoprotection is defined as the main mechanism of Sodium gualenate to protect the mucosa of the stomach and the antipeptic actions in vivo have also been shown[2]. |
| References |
| Melting Point | 98°C(lit.) |
|---|---|
| Molecular Formula | C15H17NaO3S |
| Molecular Weight | 300.348 |
| Exact Mass | 300.079620 |
| PSA | 65.58000 |
| LogP | 4.51650 |
| InChIKey | GEYJUFBPCGDENK-UHFFFAOYSA-M |
| SMILES | Cc1cc(S(=O)(=O)[O-])c2c(C)ccc(C(C)C)cc1-2.[Na+] |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RTECS | CO4826000 |
|---|---|
| HS Code | 2904100000 |
|
~% 
sodium gualenate CAS#:6223-35-4 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 21, # 19 p. 5722 - 5725 |
|
~% 
sodium gualenate CAS#:6223-35-4 |
| Literature: Bioorganic and Medicinal Chemistry Letters, , vol. 21, # 19 p. 5722 - 5725 |
| Precursor 2 | |
|---|---|
| DownStream 1 | |
| HS Code | 2904100000 |
|---|---|
| Summary | 2904100000 derivatives containing only sulpho groups, their salts and ethyl esters。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:5.5%。General tariff:30.0% |
|
Name: Antiviral activity against SARS-CoV-2 (USA-WA1/2020 strain) measured by imaging in HR...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303810
|
|
Name: Antiulcerogenic activity in Kunming mouse assessed as inhibition of ethanol-induced g...
Source: ChEMBL
Target: Mus musculus
External Id: CHEMBL1837127
|
|
Name: Antiretroviral activity against HIV-1 based virus like particles infected in U2OS cel...
Source: ChEMBL
Target: Human immunodeficiency virus 1
External Id: CHEMBL3773509
|
|
Name: Cytotoxicity against human U2OS cells assessed as cell viability at highest non-toxic...
Source: ChEMBL
Target: U2OS
External Id: CHEMBL3773508
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of VER...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4513082
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
Total 7, Current Page 1 of 1
1
| sodium 5-isopropyl-3,8-dimethylazulene-1-sulfonate |
| Gualenate de sodium |
| sodium gualenate |
| Azulene sulfonate sodium |
| 5-isopropyl-3,8-dimethyl-azulene-1-sulfonic acid,sodium-salt |
| 5-Isopropyl-3,8-dimethyl-azulen-1-sulfonsaeure,Natrium-Salz |
| Natrii gualenas |
| Sodium guiazulene sulfonate |
| Gualenato sodidico |
| Azulen SN |
| Azupromin |

 CAS#:6223-36-5
CAS#:6223-36-5