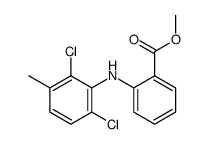
meclofenamic acid
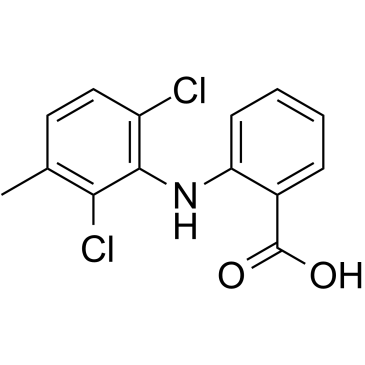
meclofenamic acid structure
|
Common Name | meclofenamic acid | ||
|---|---|---|---|---|
| CAS Number | 644-62-2 | Molecular Weight | 296.149 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 399.4±42.0 °C at 760 mmHg | |
| Molecular Formula | C14H11Cl2NO2 | Melting Point | 257-259ºC | |
| MSDS | N/A | Flash Point | 195.3±27.9 °C | |
Use of meclofenamic acidMeclofenamic acid is a nonsteroidal anti-inflammatory drug (NSAID) approved for use in arthritis (osteo and rheumatoid), analgesia (mild to moderate pain), dysmenorrhea, and heavy menstrual blood loss (menorrhagia). Meclofenamic acid is a non-selective gap-junction blocker and a highly selective inhibitor of fat mass and obesity-associated (FTO) enzyme inhibitor[1][2][3]. |
| Name | meclofenamic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Meclofenamic acid is a nonsteroidal anti-inflammatory drug (NSAID) approved for use in arthritis (osteo and rheumatoid), analgesia (mild to moderate pain), dysmenorrhea, and heavy menstrual blood loss (menorrhagia). Meclofenamic acid is a non-selective gap-junction blocker and a highly selective inhibitor of fat mass and obesity-associated (FTO) enzyme inhibitor[1][2][3]. |
|---|---|
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 399.4±42.0 °C at 760 mmHg |
| Melting Point | 257-259ºC |
| Molecular Formula | C14H11Cl2NO2 |
| Molecular Weight | 296.149 |
| Flash Point | 195.3±27.9 °C |
| Exact Mass | 295.016693 |
| PSA | 49.33000 |
| LogP | 6.67 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.663 |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| HS Code | 2922499990 |
|---|
|
~% 
meclofenamic acid CAS#:644-62-2 |
| Literature: US5462952 A1, ; US 5462952 A |
|
~% 
meclofenamic acid CAS#:644-62-2 |
| Literature: US2004/39208 A1, ; |
|
~17% 
meclofenamic acid CAS#:644-62-2 |
| Literature: Pellon, Rolando F.; Carrasco, Ramon; Marquez, Tania; Mamposo, Taimirys Tetrahedron Letters, 1997 , vol. 38, # 29 p. 5107 - 5110 |
|
~% 
meclofenamic acid CAS#:644-62-2 |
| Literature: Journal of Medicinal Chemistry, , vol. 11, # 1 p. 111 - 117 |
|
~70% 
meclofenamic acid CAS#:644-62-2 |
| Literature: Kovala-Demertzi, Dimitra; Staninska, Malgorzata; Garcia-Santos, Isabel; Castineiras, Alfonso; Demertzis, Mavroudis A. Journal of Inorganic Biochemistry, 2011 , vol. 105, # 9 p. 1187 - 1195 |
|
~% 
meclofenamic acid CAS#:644-62-2 |
| Literature: Arzneimittel-Forschung/Drug Research, , vol. 33, # 4 A p. 621 - 627 |
|
~% 
meclofenamic acid CAS#:644-62-2 |
| Literature: Crystal Growth and Design, , vol. 12, # 4 p. 2023 - 2036 |
| HS Code | 2922499990 |
|---|---|
| Summary | HS:2922499990 other amino-acids, other than those containing more than one kind of oxygen function, and their esters; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
| meclofenamate |
| 2-(2,6-dichloro-3-methylanilino)benzoic acid |
| 2-[(2,6-Dichloro-3-methylphenyl)amino]benzoic acid |
| meclofenamic acid |