Gossypin
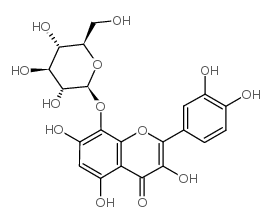
Gossypin structure
|
Common Name | Gossypin | ||
|---|---|---|---|---|
| CAS Number | 652-78-8 | Molecular Weight | 480.37600 | |
| Density | 1.883 g/cm3 | Boiling Point | 886ºC at 760 mmHg | |
| Molecular Formula | C21H20O13 | Melting Point | 229-230ºC | |
| MSDS | Chinese USA | Flash Point | 310.8ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of GossypinGossypin is a flavone isolated from Hibiscus vitifolius and has antioxidant, antiinflammatory, anticancer, anticataract, antidiabetic, analgesic and hepatoprotective activities. Gossypin inhibits NF-κB and NF-κB-regulated gene expression. Gossypin inhibits RANKL-induced osteoclastogenesis both in mouse primary bone marrow cells and RAW 264.7 cells in vitro[1][2]. |
| Name | gossypin |
|---|---|
| Synonym | More Synonyms |
| Description | Gossypin is a flavone isolated from Hibiscus vitifolius and has antioxidant, antiinflammatory, anticancer, anticataract, antidiabetic, analgesic and hepatoprotective activities. Gossypin inhibits NF-κB and NF-κB-regulated gene expression. Gossypin inhibits RANKL-induced osteoclastogenesis both in mouse primary bone marrow cells and RAW 264.7 cells in vitro[1][2]. |
|---|---|
| Related Catalog | |
| Target |
IC50: NF-κB[1] |
| References |
| Density | 1.883 g/cm3 |
|---|---|
| Boiling Point | 886ºC at 760 mmHg |
| Melting Point | 229-230ºC |
| Molecular Formula | C21H20O13 |
| Molecular Weight | 480.37600 |
| Flash Point | 310.8ºC |
| Exact Mass | 480.09000 |
| PSA | 230.74000 |
| Index of Refraction | 1.799 |
| InChIKey | SJRXVLUZMMDCNG-KKPQBLLMSA-N |
| SMILES | O=c1c(O)c(-c2ccc(O)c(O)c2)oc2c(OC3OC(CO)C(O)C(O)C3O)c(O)cc(O)c12 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 26-36-37 |
| RIDADR | NONH for all modes of transport |
|
Impact of polyphenolic components from mulberry on apoptosis of vascular smooth muscle cells.
J. Sci. Food Agric. 96 , 381-91, (2015) Previous studies have shown that mulberry polyphenolic compounds have an anti-atherosclerotic effect in rabbits. Apoptosis of vascular smooth muscle cells (VSMCs) is the key determinant of the number ... |
|
Name: Inhibition of NF-kappaB activation expressed in HCT116 cells assessed as inhibition o...
Source: ChEMBL
Target: N/A
External Id: CHEMBL1839570
|
|
Name: Aurora B Kinase Assay from Article 10.1111/cbdd.12445: "Plant-derived flavones as inh...
Source: BindingDB
Target: N/A
External Id: BindingDB_6826_1
|
|
Name: Inhibition of human liver FBP1 at 200 uM incubated for 5 mins by fluorescence method ...
Source: ChEMBL
Target: Fructose-1,6-bisphosphatase 1
External Id: CHEMBL4629675
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Binding affinity to human CDK2
Source: ChEMBL
Target: Cyclin-dependent kinase 2
External Id: CHEMBL928948
|
|
Name: Antioxidant activity assessed as DPPH free radical scavenging activity
Source: ChEMBL
Target: N/A
External Id: CHEMBL1826758
|
| Gossypetin 8-O-glucoside |
| 3,5,7,8,3',4'-hexahydroxyflavone-8-O-glucopyranoside |
| 2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-8-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one |
| 3,3',4',5,7-pentahydroxyflavone-8-O-glucoside |
| Gossypin |
| Gossypetin-8-glucoside |

