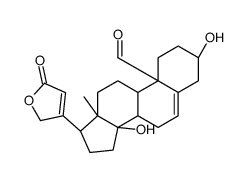
Card-20(22)-enolide,3,5,14-trihydroxy-19-oxo-, (3b,5b)-
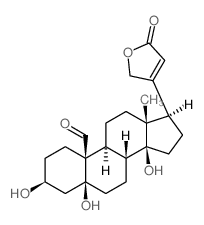
Card-20(22)-enolide,3,5,14-trihydroxy-19-oxo-, (3b,5b)- structure
|
Common Name | Card-20(22)-enolide,3,5,14-trihydroxy-19-oxo-, (3b,5b)- | ||
|---|---|---|---|---|
| CAS Number | 66-28-4 | Molecular Weight | 404.49700 | |
| Density | 1.432 g/cm3 | Boiling Point | 620.7ºC at 760 mmHg | |
| Molecular Formula | C23H32O6 | Melting Point | 169ºC | |
| MSDS | Chinese USA | Flash Point | 214.9ºC | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of Card-20(22)-enolide,3,5,14-trihydroxy-19-oxo-, (3b,5b)-Strophanthidin is a naturally available cardiac glycoside[1]. Strophanthidin 0.1 and 1 nmol/L increases and 1~100 µmol/L inhibits the Na+/K+-ATPase activities, but Strophanthidin 10 and 100 nmol/L does not affect Na+/K+-ATPase activities in cardiac sarcolemmal[2]. Strophanthidin increases both diastolic and systolic intracellular Ca2+ concentration[3]. |
| Name | strophanthidin |
|---|---|
| Synonym | More Synonyms |
| Description | Strophanthidin is a naturally available cardiac glycoside[1]. Strophanthidin 0.1 and 1 nmol/L increases and 1~100 µmol/L inhibits the Na+/K+-ATPase activities, but Strophanthidin 10 and 100 nmol/L does not affect Na+/K+-ATPase activities in cardiac sarcolemmal[2]. Strophanthidin increases both diastolic and systolic intracellular Ca2+ concentration[3]. |
|---|---|
| Related Catalog | |
| Target |
Na+/K+-ATPase[2] |
| In Vitro | Strophanthidin (0~10 μM; 24 hours; MCF-7, A549, and HepG2 cells) is effective at suppressing the growth of cancer cells and has no toxicity in normal cells[1]. Strophanthidin (0.5 to 500 µM; PBMCs) does not show significant cytotoxicity in PBMCs. Strophanthidin (2 µM; MCF-7 cells) can arrest cell cycle at the G2/M phase[1]. Strophanthidin (MCF-7, A549, and HepG2 cells) is effective at suppressing the growth of cancer cells and has no toxicity in normal cells. Strophanthidin (MCF-7, A549, and HepG2 cells) inhibits the expression of checkpoint and cyclin-dependent kinases in three cancer cells compared to untreated controls. Strophanthidin can modulate the protein localization from the nucleus to the membrane as well as to the cytoplasm. Strophanthidin is a monosaccharide cardiac glycoside with one aglycone portion and without any sugar unit. Strophanthidin induces apoptosis by the attenuation of multiple biochemical signaling pathways and by arresting cell cycle at the G2/M phase through p53-dependent and p53-independent mechanisms[1]. Cell Viability Assay[1] Cell Line: MCF-7, A549, and HepG2 cells Concentration: 0~10 μM Incubation Time: 24 hours Result: Inhibited the proliferation in three different cancer cells. |
| References |
| Density | 1.432 g/cm3 |
|---|---|
| Boiling Point | 620.7ºC at 760 mmHg |
| Melting Point | 169ºC |
| Molecular Formula | C23H32O6 |
| Molecular Weight | 404.49700 |
| Flash Point | 214.9ºC |
| Exact Mass | 404.22000 |
| PSA | 104.06000 |
| LogP | 1.89820 |
| Index of Refraction | 1.674 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H300-H310-H330 |
| Precautionary Statements | P260-P264-P280-P284-P302 + P350-P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | T+: Very toxic; |
| Risk Phrases | R26/27/28 |
| Safety Phrases | S22;S36/S37/S39;S45 |
| RIDADR | UN 2811 6 |
| RTECS | FH5425000 |
|
~82% 
Card-20(22)-eno... CAS#:66-28-4 |
| Literature: Makarevich, I. F.; Tishchenko, A. A.; Terno, I. S. Chemistry of Natural Compounds, 1991 , vol. 27, # 1 p. 53 - 55 Khimiya Prirodnykh Soedinenii, 1991 , # 1 p. 62 - 64 |
|
~%
Detail
|
| Literature: Chemistry of Natural Compounds, , vol. 22, # 2 p. 186 - 189 Khimiya Prirodnykh Soedinenii, , vol. 22, # 2 p. 201 - 204 |
|
~% 
Card-20(22)-eno... CAS#:66-28-4 |
| Literature: Gazzetta Chimica Italiana, , vol. 112, # 9/10 p. 349 - 352 |
|
Determination of strophanthidin in ingesta and plant material by LC-MS/MS.
J. Agric. Food Chem. 52 , 2174-8, (2004) An LC-MS/MS method was developed for the semiquantitative determination of strophanthidin glycosides in ingesta from animals. Strophanthidin glycosides were simultaneously extracted and hydrolyzed to ... |
|
|
Mechanistic insight into the functional and toxic effects of Strophanthidin in the failing human myocardium.
Eur. J. Heart Fail. 9(11) , 1086-94, (2007) Cardiac glycosides are characterized by a narrow therapeutic range with Ca2+-overload and arrhythmias occurring at higher concentrations. Data on cardiac glycosides in isolated failing human myocardiu... |
|
|
Summer pheasant's eye (Adonis aestivalis) poisoning in three horses.
Vet. Pathol. 41(3) , 215-20, (2004) Three horses died as a result of eating grass hay containing summer pheasant's eye (Adonis aestivalis L.), a plant containing cardenolides similar to oleander and foxglove. A 9-year-old thoroughbred g... |
| Convallatoxigenin |
| Corchorgenin |
| Corchsularin |
| Strophanthidin K |
| MFCD00046266 |
| EINECS 200-626-6 |
| (3S,5S,8R,9S,10S,13R,14S,17R)-3,5,14-trihydroxy-13-methyl-17-(5-oxo-2H-furan-3-yl)-2,3,4,6,7,8,9,11,12,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-10-carbaldehyde |
| k-Strophanthidin |
| Strophanthidine |
| Erysimupicrone |
| Corchorin |
| Corchoside A aglycon |

 CAS#:508-75-8
CAS#:508-75-8![(3S,5S,10S,13R,14S,17R)-5,14-dihydroxy-13-methyl-17-(5-oxo-2H-furan-3-yl)-3-[(2S,5R)-3,4,5-trihydroxyoxan-2-yl]oxy-2,3,4,6,7,8,9,11,12,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-10-carbaldehyde structure](https://www.chemsrc.com/caspic/116/15596-26-6.png) CAS#:15596-26-6
CAS#:15596-26-6![[10-formyl-5,14-dihydroxy-13-methyl-17-(5-oxo-2H-furan-3-yl)-2,3,4,6,7,8,9,11,12,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-yl] 2-iodoacetate structure](https://www.chemsrc.com/caspic/426/4956-17-6.png) CAS#:4956-17-6
CAS#:4956-17-6 CAS#:4956-18-7
CAS#:4956-18-7
