Norethynodrel
Modify Date: 2025-08-21 10:45:18
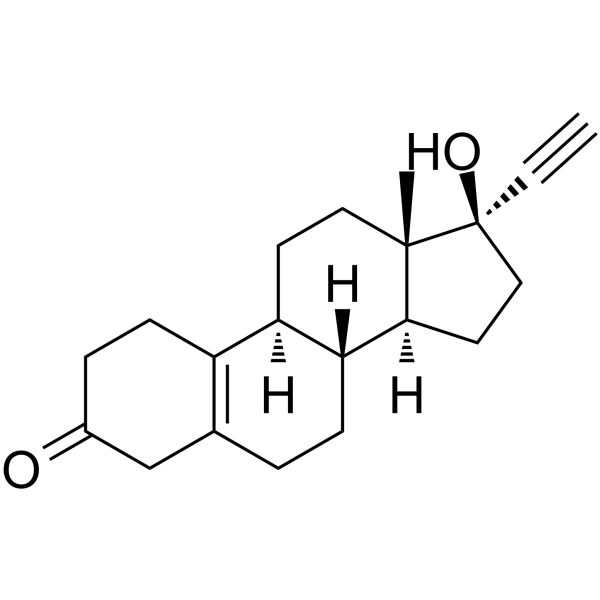
Norethynodrel structure
|
Common Name | Norethynodrel | ||
|---|---|---|---|---|
| CAS Number | 68-23-5 | Molecular Weight | 298.41900 | |
| Density | 1.15g/cm3 | Boiling Point | 441.6ºC at 760 mmHg | |
| Molecular Formula | C20H26O2 | Melting Point | 169-170ºC | |
| MSDS | N/A | Flash Point | 188.2ºC | |
Use of NorethynodrelNorethynodrel (Enidre) is a 19-nortestosterone derivative to be used as a progestagen. Norethynodrel can be used for researching inhibition of ovulation[1]. |
| Name | (8R,9S,13S,14S,17R)-17-ethynyl-17-hydroxy-13-methyl-1,2,4,6,7,8,9,11,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-3-one |
|---|---|
| Synonym | More Synonyms |
| Description | Norethynodrel (Enidre) is a 19-nortestosterone derivative to be used as a progestagen. Norethynodrel can be used for researching inhibition of ovulation[1]. |
|---|---|
| Related Catalog | |
| References |
[1]. D F HAWKINS, et al. 2 - The Clinical Pharmacology of Oestrogens and Progestagens. 1979, |
| Density | 1.15g/cm3 |
|---|---|
| Boiling Point | 441.6ºC at 760 mmHg |
| Melting Point | 169-170ºC |
| Molecular Formula | C20H26O2 |
| Molecular Weight | 298.41900 |
| Flash Point | 188.2ºC |
| Exact Mass | 298.19300 |
| PSA | 37.30000 |
| LogP | 3.63660 |
| Index of Refraction | 1.577 |
| InChIKey | ICTXHFFSOAJUMG-SLHNCBLASA-N |
| SMILES | C#CC1(O)CCC2C3CCC4=C(CCC(=O)C4)C3CCC21C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Hazard Codes | T |
|---|---|
| Risk Phrases | 45-61 |
| Safety Phrases | 53-22-36/37/39-45 |
| WGK Germany | 3 |
| RTECS | RC8980000 |
|
~% 
Norethynodrel CAS#:68-23-5 |
| Literature: US2691028 , ; |
| Precursor 0 | |
|---|---|
| DownStream 2 | |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: MITF Measured in Cell-Based System Using Plate Reader - 2084-01_Activator_SinglePoint...
Source: Broad Institute
Target: N/A
External Id: 2084-01_Activator_SinglePoint_HTS_Activity
|
|
Name: Counterscreen for inhibitors of the fructose-bisphosphate aldolase (FBA) of M. tuberc...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: GDH-TPI_INH_ABS_1536_1X%INH CSRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: Inhibition of cell viability relative to arbidol control (inhibition index > 1 indica...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303819
|
Total 260, Current Page 1 of 26
1
2
3
4
5
| SC-4642 |
| Enovid |
| Norethynodrel [Progestins] |
| norethynodrel |
| Noretynodrel |
| Noretinodrel [DCIT] |
| 19-Norethynodrel |
| Norethinodrel |
| Lynestrol |

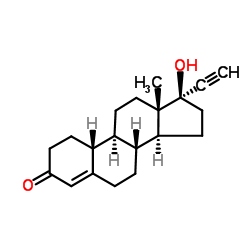 CAS#:68-22-4
CAS#:68-22-4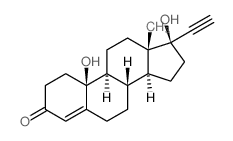 CAS#:1236-00-6
CAS#:1236-00-6