Quinethazone
Modify Date: 2025-08-26 19:15:31
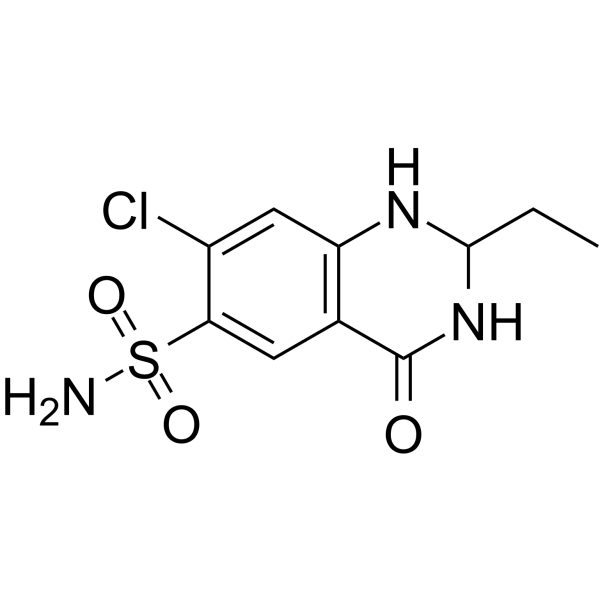
Quinethazone structure
|
Common Name | Quinethazone | ||
|---|---|---|---|---|
| CAS Number | 73-49-4 | Molecular Weight | 289.739 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C10H12ClN3O3S | Melting Point | 250 - 252ºC | |
| MSDS | N/A | Flash Point | N/A | |
Use of QuinethazoneQuinethazone is an orally active diuretic agent and is also a weak inhibitor of carbonic anhydrase. Quinethazone can be used for hypertension research[1][2]. |
| Name | 7-chloro-2-ethyl-4-oxo-2,3-dihydro-1H-quinazoline-6-sulfonamide |
|---|---|
| Synonym | More Synonyms |
| Description | Quinethazone is an orally active diuretic agent and is also a weak inhibitor of carbonic anhydrase. Quinethazone can be used for hypertension research[1][2]. |
|---|---|
| Related Catalog | |
| In Vivo | Quinethazone (50 mg/kg; p.o.; daily for 2 weeks) lowers blood pressure of renal hypertensive rats in association with a decrease in potassium but no change in water or sodium contents of aorta wall[1]. Animal Model: Five- to 6-week-old Long Evans male rats, hypertension model[1] Dosage: 50 mg/kg Administration: Oral, daily for 2 weeks Result: Had no significant effect on sodium or water distribution in arterial tissue of hypertensive rats, but decreased approximate 20% intra-cellular content of potassium |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Melting Point | 250 - 252ºC |
| Molecular Formula | C10H12ClN3O3S |
| Molecular Weight | 289.739 |
| Exact Mass | 289.028778 |
| PSA | 109.67000 |
| LogP | 0.49 |
| Index of Refraction | 1.582 |
| InChIKey | AGMMTXLNIQSRCG-UHFFFAOYSA-N |
| SMILES | CCC1NC(=O)c2cc(S(N)(=O)=O)c(Cl)cc2N1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | NONH for all modes of transport |
|---|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
Total 467, Current Page 1 of 47
1
2
3
4
5
| Quimethazin |
| Quinethazonum |
| Hydromox |
| Quinetazona |
| Quinethazon |
| Chinethazonum |
| 7-Chlor-6-sulfamoyl-2-aethyl-4-oxo-1,2,3,4-tetrahydro-chinazolin |
| UNII:455E0S048W |
| 7-Chlor-2-aethyl-6-sulfamoyl-1,2,3,4-tetrahydro-4-chinazolinon |
| quinethazone |
| 7-chloro-2-ethyl-4-oxo-1,2,3,4-tetrahydro-quinazoline-6-sulfonic acid amide |
| 7-chloro-2-ethyl-1,2,3,4-tetrahydro-4-oxo-6-quinazolinesulfonamide |
| Aquamox |
| 7-Chloro-2-ethyl-4-oxo-1,2,3,4-tetrahydro-6-quinazolinesulfonamide |
| 7-Chloro-2-ethyl-4-oxo-1,2,3,4-tetrahydroquinazoline-6-sulfonamide |
| Idrokin |